Research Articles
From Formality to Foundation: The Modern Evolution of Informed Consent Regulations in Clinical Research
This article traces the significant evolution of informed consent regulations, moving beyond a one-time signature to an ongoing, ethical dialogue.
The Belmont Report in Practice: Applying Ethical Principles and Informed Consent in Modern Clinical Research
This article provides a comprehensive guide to the Belmont Report's ethical principles—Respect for Persons, Beneficence, and Justice—and their critical application in contemporary biomedical research.
From Paternalism to Patient Autonomy: The Evolving History and Future of Informed Consent in Clinical Research
This article traces the complex evolution of informed consent from its early 20th-century legal foundations to the sophisticated ethical and regulatory framework of 2025.
The Nuremberg Code in Modern Research: Applying Informed Consent Principles in Drug Development
This article provides a comprehensive analysis of the Nuremberg Code's informed consent principles for researchers, scientists, and drug development professionals.
Integrating Theological Bioethics in Clinical Practice: A Framework for Humanized Patient Care and Ethical Drug Development
This article addresses the growing need to validate theological and religious perspectives within secular bioethics, particularly for researchers and drug development professionals navigating complex ethical landscapes.
Validating Bioethics Text Classification Models: A Framework for Ethical AI in Healthcare Research
The integration of Large Language Models (LLMs) and other AI systems for classifying bioethics-related text in healthcare presents both unprecedented opportunities and profound ethical challenges.
Rigorous and Ethical Synthesis: A Comprehensive Guide to Evaluating Systematic Review Methods in Bioethics
This article provides a comprehensive framework for researchers, scientists, and drug development professionals to critically evaluate systematic review methodologies within the unique domain of bioethics.
Navigating the Methodological Landscape: A Comparative Study of Empirical Bioethics Approaches for Biomedical Research
This article provides a systematic comparison of empirical bioethics methodologies, tailored for researchers, scientists, and drug development professionals.
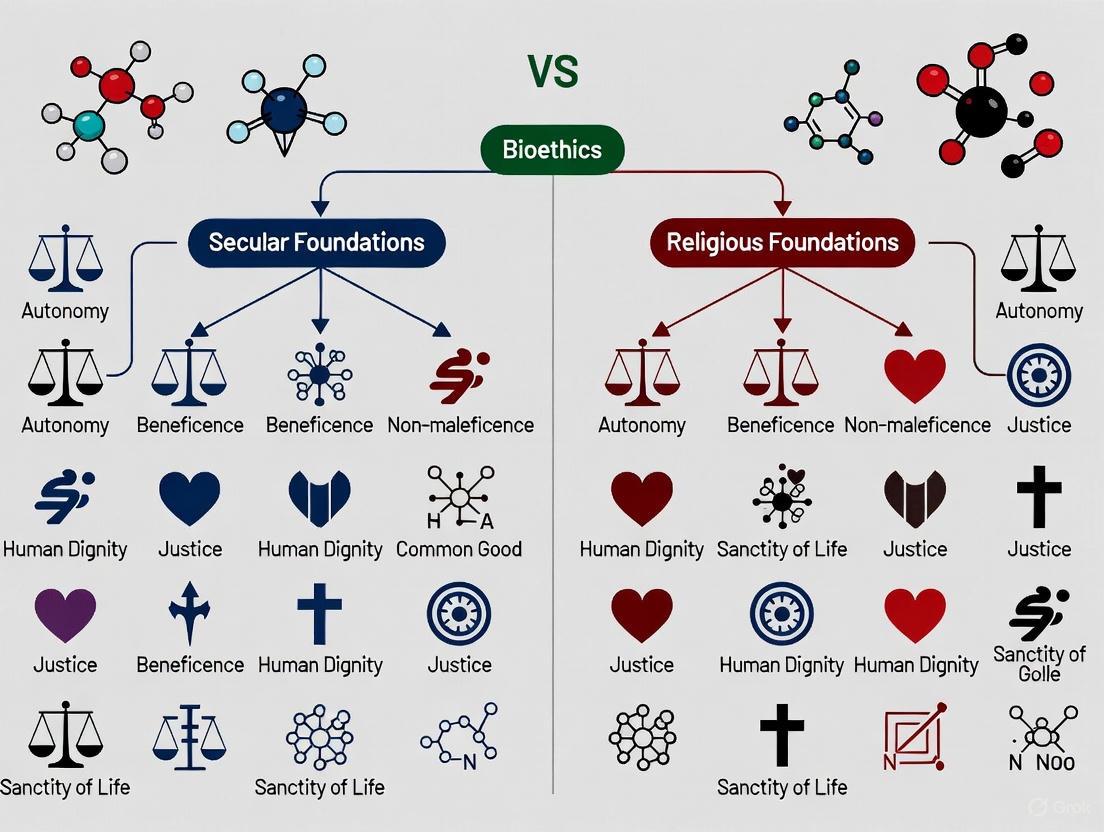
Secular vs. Religious Bioethics: Foundations, Applications, and Resolutions for Biomedical Research
This article provides a comparative analysis of the foundational principles and methodological applications of secular and religious bioethical traditions.
Empirical Bioethics Methodology: Assessing Rigor, Navigating Standards, and Implementing Best Practices
This article provides a comprehensive framework for assessing methodological rigor in empirical bioethics, tailored for researchers, scientists, and drug development professionals.