Research Articles
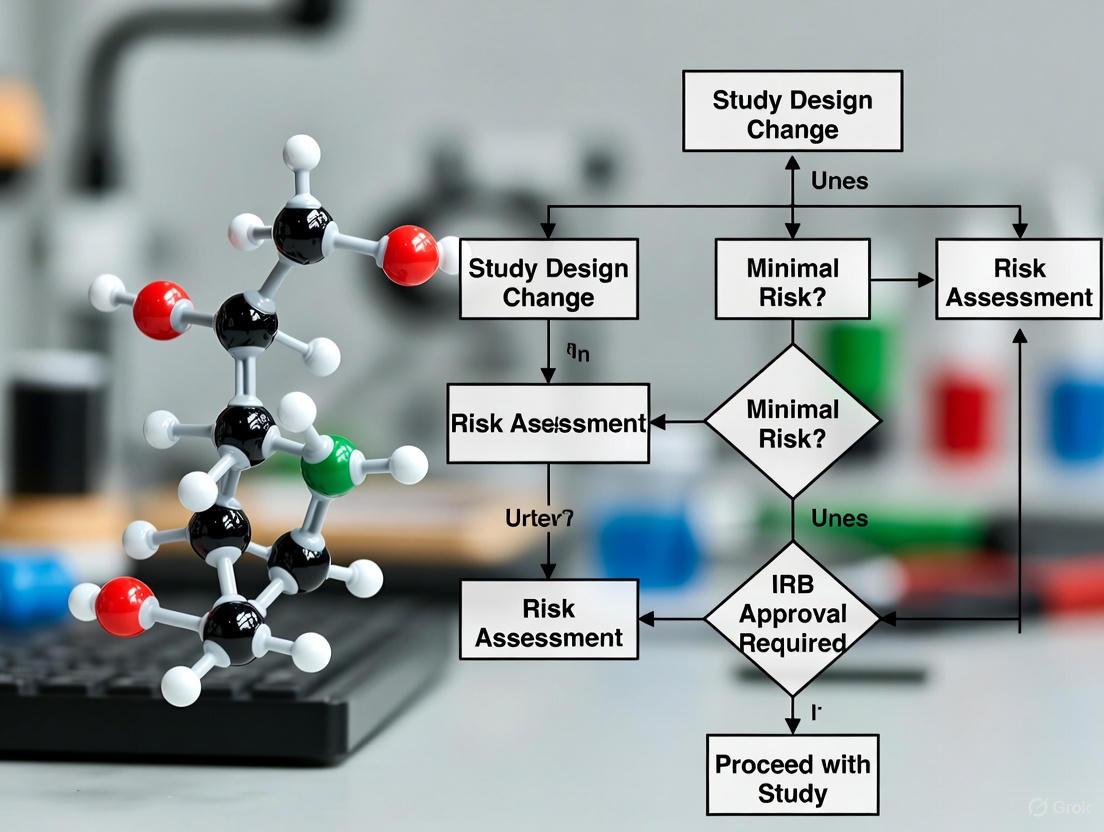
When Is IRB Approval Required for a Study Change? A Guide for Researchers
This article provides a comprehensive guide for researchers and drug development professionals on navigating Institutional Review Board (IRB) amendments.
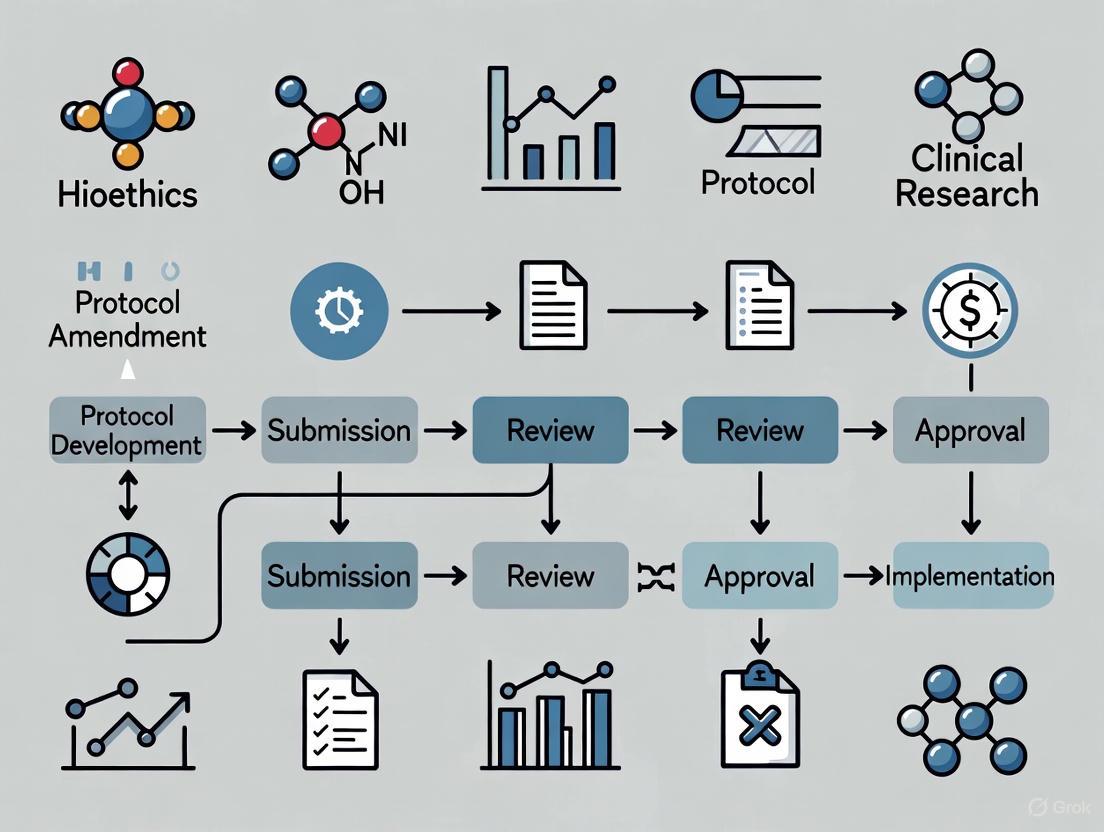
Protocol Amendments in Clinical Research: A Comprehensive Guide to Management, Costs, and Compliance
This article provides a complete overview of protocol amendments in clinical trials for drug development professionals and researchers.
Ethical Consent in Clinical Trials: A Comparative Analysis of Processes for Vulnerable Populations
This article provides a comprehensive analysis of informed consent processes for vulnerable populations in clinical research.
Evaluating Key Information Section Impact on Understanding: A Strategic Guide for Clinical Research Professionals
This article provides a comprehensive framework for researchers, scientists, and drug development professionals to evaluate and enhance the impact of Key Information sections in informed consent forms.
Informed Consent Comprehension Rates Across Medical Specialties: A Systematic Analysis for Clinical Research Professionals
This comprehensive review examines the critical challenge of variable informed consent comprehension rates across medical specialties.
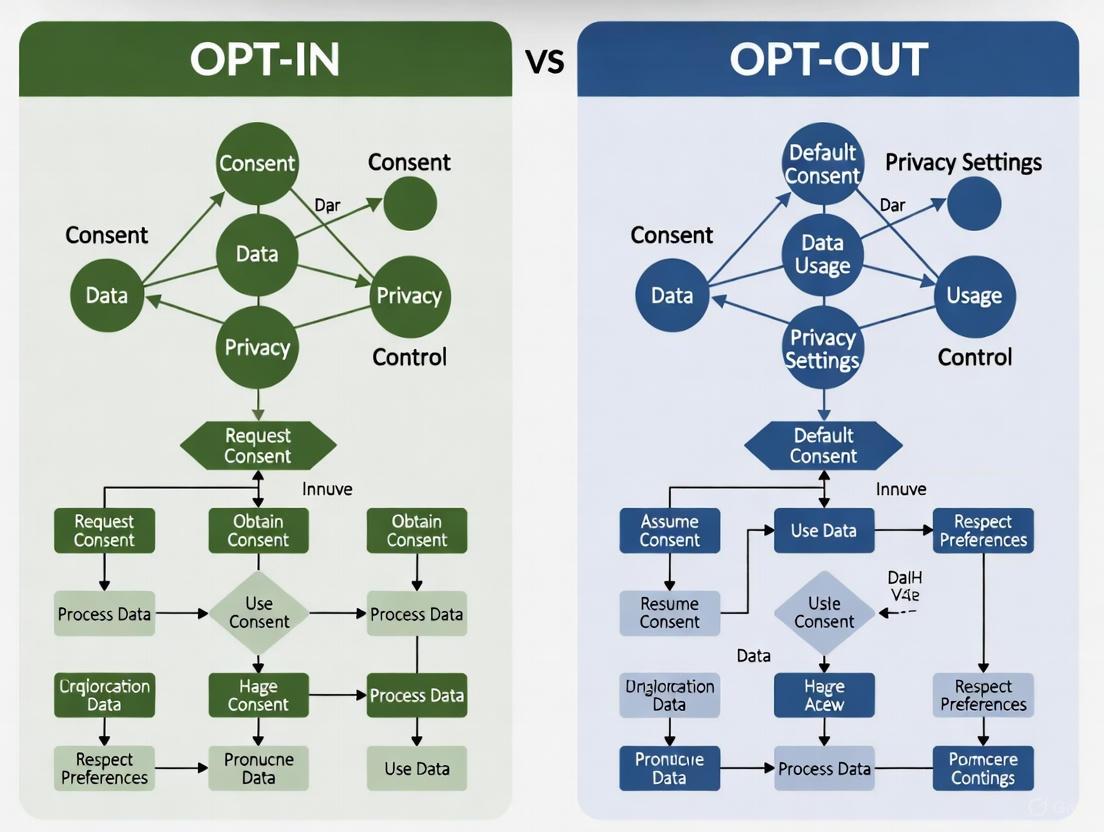
Opt-In vs Opt-Out Consent: A Strategic Guide for Biomedical Research and Drug Development
This article provides a comprehensive comparison of opt-in and opt-out consent models, tailored for researchers, scientists, and drug development professionals.
Global Informed Consent Guidelines: A Comparative Analysis for International Research and Drug Development
This article provides a comprehensive comparison of international informed consent guidelines, addressing critical needs for researchers and drug development professionals engaged in multi-country studies.
Validated Tools for Assessing Informed Consent Understanding: A Comprehensive Guide for Clinical Researchers
This article provides clinical researchers and drug development professionals with a comprehensive overview of validated tools and methodologies for assessing participant understanding in the informed consent process.
Beyond Paper Consent: A Comparative Analysis of Modern Presentation Methods for Enhanced Clinical Trial Participation
This article provides a comprehensive analysis for researchers and drug development professionals on the comparative effectiveness of various informed consent presentation methods.
Beyond the Signature: Empirical Evidence on Patient Comprehension in Informed Consent and Pathways to Improvement
This article synthesizes empirical findings on a critical challenge in clinical research and practice: the widespread deficiency in patient understanding of informed consent forms.