The $1 Million Amendment: A Strategic Cost Comparison of Avoidable vs. Necessary Clinical Trial Changes
This article provides clinical researchers and drug development professionals with a data-driven framework to analyze, manage, and reduce clinical trial protocol amendments.
The $1 Million Amendment: A Strategic Cost Comparison of Avoidable vs. Necessary Clinical Trial Changes
Abstract
This article provides clinical researchers and drug development professionals with a data-driven framework to analyze, manage, and reduce clinical trial protocol amendments. Drawing on the latest industry benchmarks, we break down the direct and indirect costs of both necessary and avoidable amendments, explore root causes—from flawed eligibility criteria to rushed planning—and present actionable strategies for prevention. By comparing the financial and operational impact of different amendment types and showcasing effective mitigation tactics, this guide aims to equip teams with the knowledge to enhance trial efficiency, control budgets, and accelerate the delivery of new therapies to market.
Understanding Protocol Amendments: Prevalence, Costs, and Impact on Trial Success
In clinical research, a protocol amendment is defined as any change to a clinical trial protocol that requires internal approval followed by approval from an Institutional Review Board (IRB) or regulatory authority [1]. These modifications are a fundamental aspect of trial management but carry significant operational and financial implications. Recent benchmarks reveal that 76% of Phase I-IV clinical trials now require at least one protocol amendment, a substantial increase from 57% in 2015 [2] [3]. The financial impact is equally striking, with direct costs ranging from $141,000 to $535,000 per amendment, not accounting for indirect expenses from delayed timelines and site disruptions [2].
This guide provides an objective comparison between necessary and avoidable amendments, framing the analysis within a broader cost-comparison thesis to equip researchers and drug development professionals with evidence-based strategies for optimal protocol management.
Table 1: Protocol Amendment Benchmarks and Financial Impact (2022-2024 Data)
| Metric | Phase I | Phase II | Phase III | Phase IV | Source |
|---|---|---|---|---|---|
| Prevalence of Protocols with ≥1 Amendment | Increased (Specific % N/A) | 89% | Increased (Specific % N/A) | Increased (Specific % N/A) | [4] |
| Mean Number of Amendments per Protocol | High Increase | High Increase | [3] | ||
| Direct Cost per Amendment | $141,000 (Median) | $535,000 (Median) | [2] [5] | ||
| Average Implementation Timeline | 260 days (from identification to final approval) | [2] [3] |
Table 2: Categorization and Causes of Protocol Amendments
| Category | Definition | Common Examples | Proportion of Amendments |
|---|---|---|---|
| Necessary Amendments | Changes driven by external factors or critical needs for trial validity/safety. | - New safety information [1]- Regulatory agency requests [3] [1]- New scientific findings [2] | 66-77% [3] [1] |
| Avoidable Amendments | Changes resulting from internal protocol design flaws or poor planning. | - Protocol design flaws/inconsistencies [1] [5]- Eligibility criteria adjustments [2]- Changes to protocol title/administrative info [2] | 23-34% [2] [1] |
Comparative Analysis: Necessary vs. Avoidable Amendments
Necessary Amendments: Unavoidable Solutions
Necessary amendments are primarily reactive, driven by external factors or critical needs that arise during the trial's conduct. The top cause for amendments, accounting for nearly 20% of changes, is new safety information emerging from the ongoing study or external sources [1]. These amendments are crucial for ensuring participant safety and may include new adverse event monitoring requirements [2].
Another significant driver is regulatory agency requests, which constitute 18.6% of amendments [1]. These often involve compliance with updated FDA/EMA guidance [2]. A 2024 study confirmed that regulatory requests, along with changes to the study strategy, remain top reasons for amending a protocol, with 77% of all amendments deemed unavoidable [3]. These changes reflect the dynamic nature of clinical science and regulatory landscapes, where adaptations are essential for maintaining trial relevance and compliance.
Avoidable Amendments: The Preventable Cost
In contrast, avoidable amendments often stem from internal planning deficiencies. A consistent finding across studies is that protocol design flaws and inconsistencies cause approximately 11% of all amendments [1] [5]. These include issues such as unclear eligibility criteria, impractical assessment schedules, and procedural inconsistencies that could have been identified and corrected during protocol development.
Recruitment challenges constitute another major category of avoidable amendments, causing 9% of total amendments [1]. A 2023 study of NHS trials identified "to achieve the trial's recruitment target" as the single most common reason for amendments, with "addition of sites" being the most frequent change [6]. This suggests insufficient feasibility assessment during planning phases. Minor administrative changes, such as altering the protocol title or contact information, also contribute to avoidable amendments. While seemingly insignificant, these changes trigger full regulatory review processes, creating unnecessary administrative burdens [2].
Experimental Protocols for Amendment Research
Methodology 1: Content Analysis of Amendment Documents
Research by Joshi et al. (2023) established a robust methodology for analyzing amendment patterns through systematic content analysis [6]:
- Data Collection: Researchers collected 242 approved amendments from 53 clinical research studies sponsored by a University Hospital NHS Trust over an 11-year period (2009-2020)
- Coding Framework: Using conventional content analysis, researchers developed categories directly from text data rather than pre-defined classifications
- Unit of Analysis: Individual amendment "Changes" and "Reasons" were used as recording units, with NVivo 12 Plus software organizing codes into content-related categories
- Validation: 5% of the sample (n=12) was randomly selected for independent coding by a second researcher to ensure reproducibility of the coding framework
This methodology enables systematic identification of amendment patterns and root causes across large datasets of historical trial documents.
Methodology 2: Stakeholder Interviews for Root Cause Analysis
The same study employed semi-structured interviews to explore the underlying reasons for amendments [6]:
- Participant Selection: Researchers invited approximately 150 research staff with experience in amendment development, review, or implementation
- Qualitative Analysis: Eleven semi-structured interviews were conducted, transcribed verbatim, and analyzed thematically using the Framework approach
- Triangulation: Interview findings were integrated with content analysis results to provide depth and context to the quantitative data
This mixed-methods approach provides both statistical patterns and deeper insights into the operational challenges driving amendments.
Strategic Framework for Amendment Management
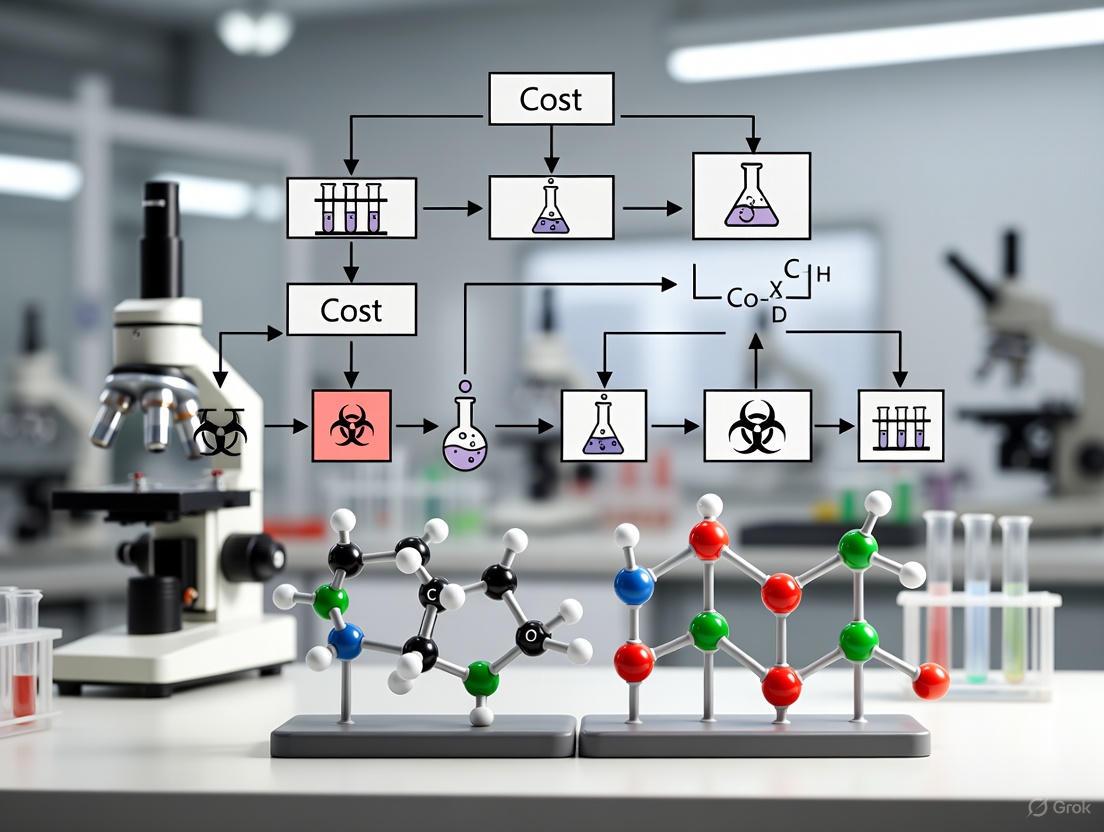
The following workflow outlines a strategic approach to protocol amendments, from identification through implementation, helping teams distinguish between necessary and avoidable changes while minimizing disruption.
Research Reagent Solutions: Tools for Amendment Reduction
Table 3: Essential Resources for Optimal Protocol Design
| Research Tool/Solution | Function | Application Context |
|---|---|---|
| Multidisciplinary Review Committees | Provides balanced assessment of scientific validity and operational feasibility during protocol design [7]. | Early protocol development phase to stress-test concepts and identify potential design flaws. |
| Patient Advisory Boards | Incorporates patient perspective on burden, visit frequency, and procedures to improve recruitment and retention [2] [7]. | Protocol finalization to assess patient-centricity and real-world feasibility. |
| Standard of Care (SoC) Databases | Provides insights into local treatment pathways and healthcare practices to align trial design with real-world settings [5]. | Feasibility assessment and country/site selection to ensure protocol alignment with local practices. |
| Protocol Optimization Worksheets | Structured checklists to quantify protocol quality and complexity before finalization [7]. | Protocol finalization to objectively assess complexity and identify unnecessary procedures. |
| AI-Powered Design Tools | Leverages machine learning to predict design flaws and optimize eligibility criteria [7] [8]. | Early protocol development to model different design scenarios and predict operational impact. |
Protocol amendments represent a significant cost center in clinical development, with nearly one-quarter to one-third being potentially avoidable through improved planning and design practices [2] [1]. The distinction between necessary and avoidable amendments is not merely academic—it has direct financial consequences, with avoidable amendments costing the industry approximately $2 billion annually in direct costs alone [1].
Successful amendment management requires a balanced approach: embracing necessary changes that enhance trial safety and scientific validity while systematically reducing avoidable amendments through robust protocol development processes. Organizations that master this balance through early stakeholder engagement, feasibility assessment, and strategic planning stand to gain significant advantages in trial efficiency, cost management, and ultimately, faster delivery of new treatments to patients.
In the landscape of clinical development, protocol amendments have become a pervasive and costly reality. Recent data reveals a significant escalation in both the frequency and financial impact of changes to clinical trial protocols, presenting a substantial challenge for research efficiency and budget management. For drug development professionals, understanding the current benchmarks of avoidable versus necessary amendments is crucial for implementing effective cost-containment strategies. This analysis examines the 2024 data on amendment trends, providing a detailed comparison of their operational and financial implications to guide more strategic protocol planning and execution.
The rising tide of amendments reflects fundamental changes in clinical research complexity, particularly in oncology and rare disease studies. A groundbreaking 2024 study from the Tufts Center for the Study of Drug Development (CSDD) establishes new benchmarks, indicating that 76% of Phase I-IV trials now require amendments, a dramatic increase from 57% in 2015 [2]. This trend is especially pronounced in specific therapeutic areas, with research showing that 90% of oncology trials require at least one amendment [2]. This escalation stems from multiple factors, including evolving scientific understanding, increasing regulatory requirements, and the inherent complexity of modern trial designs.
Quantitative Analysis: Amendment Prevalence and Cost Structures
The financial implications of protocol amendments are substantial, with each change triggering a cascading effect across multiple trial operations. The direct and indirect costs associated with these amendments have created a significant burden on clinical development budgets.
Table 1: Clinical Trial Amendment Benchmarks (2024 Data)
| Metric | 2024 Benchmark | Change from 2015 | Key Influencing Factors |
|---|---|---|---|
| Overall Amendment Rate | 76% of Phase I-IV trials [2] | Increase from 57% [2] | Increasing trial complexity, regulatory requirements, novel endpoints |
| Oncology Trial Amendment Rate | ~90% of trials [2] | Not specified | Complex biomarkers, novel endpoints, combination therapies |
| Average Implementation Timeline | 260 days [2] | Not specified | IRB reviews, site retraining, contract updates, database changes |
| Direct Cost per Amendment | $141,000 - $535,000 [2] | Not specified | Phase of trial, scope of changes, number of sites affected |
| Potentially Avoidable Amendments | 23% [2] | Not specified | Protocol title changes, minor eligibility adjustments, assessment schedule modifications |
Beyond the direct costs outlined in Table 1, amendments trigger substantial indirect expenses through operational disruptions. The implementation of amendments now averages 260 days, with sites operating under different protocol versions for an average of 215 days, creating significant compliance risks [2]. These timeline extensions delay crucial medical innovations and increase the operational burden on research sites. Additionally, a study published in Pharmacology and Therapeutics estimated the average patient cost in a clinical trial at $41,413, meaning amendments that extend trial duration or require additional patient visits can dramatically increase these costs [9].
Categorizing Amendments: Necessary vs. Avoidable
Understanding the distinction between necessary and avoidable amendments enables sponsors to make more informed decisions and prioritize prevention strategies where they will have the greatest impact.
Necessary Amendments
Essential amendments typically fall into three categories:
- Safety-Driven Changes: Implementation of new adverse event monitoring requirements based on emerging data [2]
- Regulatory-Required Adjustments: Compliance with updated FDA/EMA guidance issued during trial conduct [2]
- New Scientific Findings: Incorporation of biomarker-driven stratification based on newly published evidence [2]
These amendments are fundamentally unavoidable and often enhance patient safety or trial scientific validity.
Avoidable Amendments
Research indicates that approximately 23% of amendments are potentially avoidable, representing a significant opportunity for cost containment [2]. Common avoidable amendments include:
- Protocol Title Changes: Altering the study title creates unnecessary administrative burden and requires updates to all regulatory filings [2]
- Minor Eligibility Adjustments: Small changes in inclusion/exclusion criteria trigger the need for revised 1572's and consents, requiring patient reconsent and adding regulatory complexity [2]
- Assessment Schedule Modifications: Moving a single assessment can alter site budget agreements and require updates and revalidation of electronic data capture (EDC) systems [2]
These avoidable amendments frequently occur because companies fail to fully evaluate the downstream impact before initiating changes, allowing small modifications to cascade into major logistical and financial burdens.
Experimental Protocols and Methodologies
The methodology for classifying and analyzing amendment data follows rigorous research protocols essential for accurate benchmarking and trend analysis.
Amendment Classification Methodology
The Tufts CSDD study, which provides the foundational 2024 data, employs a systematic classification framework:
- Amendment Identification: Researchers collect protocol change data from participating pharmaceutical companies, contract research organizations (CROs), and investigative sites
- Categorization Protocol: Each amendment is coded according to:
- Primary driver (safety, regulatory, operational, scientific)
- Phase of occurrence
- Therapeutic area
- Scope of changes required
- Impact Assessment: Researchers quantify both direct costs (IRB fees, contract updates) and indirect costs (timeline delays, resource diversion)
- Avoidability Analysis: A panel of experts reviews each amendment against predefined criteria to determine whether better planning could have prevented it
This structured approach enables consistent benchmarking across the industry and year-over-year trend analysis.
Cost Calculation Methodology
The financial impact analysis follows a comprehensive approach:
- Direct Cost Tracking: Documentation of all expenses associated with amendment implementation, including:
- IRB review fees and administrative costs
- Regulatory submission preparation
- Site contract and budget renegotiations
- Database updates and validation
- Documentation and training material revisions
- Indirect Cost Estimation: Calculation of expenses related to:
- Timeline extensions (additional months of site monitoring, management overhead)
- Resource diversion (clinical, regulatory, and data management staff time)
- Opportunity costs (delayed product launch, extended patent life)
- Cross-Industry Aggregation: Normalization and aggregation of data across multiple organizations to establish industry benchmarks
This methodology provides the comprehensive cost ranges cited in Table 1, which represent both immediate implementation expenses and broader operational impacts.
Visualizing the Amendment Decision Pathway
The following diagram illustrates the clinical trial amendment decision pathway, categorizing amendments by type and outlining their distinct impacts on trial execution.
Figure 1: Clinical Trial Amendment Decision Pathway
The Researcher's Toolkit: Essential Solutions for Protocol Optimization
Implementing the right tools and strategies can significantly reduce amendment frequency and improve protocol quality. The following table details key research solutions that address common amendment drivers.
Table 2: Research Reagent Solutions for Protocol Optimization
| Solution Category | Specific Tools/Approaches | Function in Amendment Prevention |
|---|---|---|
| Stakeholder Engagement Platforms | Patient advisory boards, Site feasibility surveys | Identifies operational barriers and patient burden early in design phase [2] |
| Data Analytics & Feasibility | TA Scan, Competitive trial analysis | Provides pre-existing data for feasibility assessments, enrollment benchmarking [9] |
| Protocol Authoring Tools | Structured template libraries, Electronic protocol systems | Standardizes protocol elements and reduces ambiguity [2] |
| Amendment Management Systems | Dedicated amendment teams, Decision framework tools | Provides structured approach to evaluate necessity and impact of changes [2] |
| Clinical Data Management | eClinical suites, Automated data validation | Streamlines data collection and identifies issues early [9] |
These solutions directly target the root causes of avoidable amendments. For instance, comprehensive data analytics tools like TA Scan gather and aggregate data from diverse registries and online repositories, providing a wealth of pre-existing information for feasibility assessments and enrollment benchmarking [9]. This addresses one of the most common drivers of amendments - poor initial feasibility assessment. Similarly, engaging key stakeholders early in protocol design, including regulatory experts, site staff, and patient advisors, helps identify potential operational challenges before the protocol is finalized [2].
The 2024 data presents a clear call to action for the clinical research industry. With amendment rates rising to 76% of trials and avoidable amendments consuming substantial resources, organizations must prioritize strategic protocol planning and implementation. The difference between a well-planned amendment and an avoidable one can mean millions of dollars in additional trial costs and significant delays in delivering new treatments to patients [2].
Organizations that master this balance stand to gain significant advantages through improved trial efficiency and reduced operational costs. Successful strategies include engaging key stakeholders early in protocol development, bundling amendments strategically when possible, establishing dedicated amendment teams for consistency, and implementing clear communication frameworks to ensure smooth amendment adoption [2]. Additionally, leveraging digital tools for clinical data management can help streamline operations and reduce avoidable delays and costs [9]. By adopting these approaches, drug development professionals can navigate the rising tide of amendments more effectively, containing costs without compromising scientific integrity or patient safety.
In the high-stakes realm of clinical research, a comprehensive understanding of cost structures is not merely an accounting exercise—it is a fundamental component of strategic trial management and financial sustainability. For drug development professionals, the ability to dissect and analyze direct, indirect, and hidden expenses is critical for maintaining budgetary control and operational efficiency. This is particularly true when evaluating protocol amendments, a near-ubiquitous occurrence in clinical research that can dramatically alter a trial's financial trajectory. A recent study reveals that 76% of Phase I-IV trials now require at least one protocol amendment, a significant increase from 57% in 2015 [2]. Each amendment carries a direct price tag ranging from $141,000 to $535,000, but this figure represents only the tip of the financial iceberg, as it excludes substantial indirect costs and hidden expenses that can multiply the total economic impact [2]. This guide provides a structured framework for comparing avoidable versus necessary amendments, offering researchers and scientists the data, methodologies, and tools to make informed financial decisions.
The Fundamental Cost Categories in Clinical Research
In clinical research, all expenses can be categorized into three distinct types: direct, indirect, and hidden costs. Understanding the differences between them is the first step toward effective cost management.
Direct Costs are expenses that can be specifically and exclusively traced to a clinical trial or a specific cost object like a product or project. These are the tangible, easily identifiable costs of conducting the research itself [10] [11] [12].
Indirect Costs are expenses that support the overall clinical research operation but cannot be tied to a single trial or project. These are the overhead costs necessary for maintaining the research infrastructure [10] [11] [13].
Hidden Costs are unanticipated or less visible expenses that arise as a consequence of changes or disruptions in the trial process. Unlike direct and indirect costs, they are not planned for and are often realized only after an event like a protocol amendment occurs [2].
Table: Comparison of Fundamental Cost Categories
| Cost Category | Definition | Traceability | Examples in Clinical Trials |
|---|---|---|---|
| Direct Costs | Expenses directly tied to a specific clinical trial or project [10] [12]. | Easily traceable to a specific cost object [10] [11]. | Clinical protocol procedures, investigator grants, central laboratory costs, clinical trial technology, patient recruitment costs, protocol-specific imaging [2]. |
| Indirect Costs | Overhead expenses necessary for general operations but not tied to one trial [10] [13]. | Shared across multiple projects/departments; requires allocation [10] [12]. | Rent for facility space, salaries of HR/legal staff, utilities, general office supplies, depreciation of shared equipment [10] [11]. |
| Hidden Costs | Unanticipated costs from operational inefficiencies or disruptions [2]. | Difficult to foresee and quantify; often a cascading effect. | Patient re-consent delays, site re-training, data management revisions, statistical plan rework, extended project timelines [2]. |
Quantitative Analysis of Protocol Amendment Costs
Protocol amendments are a primary driver of cost escalation in clinical trials. A granular breakdown of their financial impact is essential for distinguishing between the costs of necessary and avoidable changes.
The Financial Cascade of a Single Amendment
A protocol amendment triggers a cascade of activities across multiple functional areas. The initial cost of the change itself is often dwarfed by the downstream operational impacts. Research indicates that the implementation of amendments now averages 260 days, with sites often operating under different protocol versions for an average of 215 days, creating significant compliance risks and operational friction [2].
Table: Cost Breakdown of a Typical Protocol Amendment
| Cost Category | Specific Cost Driver | Financial Impact / Range | Operational Consequences |
|---|---|---|---|
| Direct Costs | IRB/Regulatory Submission Fees | $XX,XXX | Adds weeks to timelines; requires formal review and approval [2]. |
| Site Budget Re-negotiations | $XX,XXX | Increases legal and administrative costs; delays site activation and adoption [2]. | |
| Data Management & EDC System Updates | $XX,XXX | Requires reprogramming, validation, and database updates [2]. | |
| Indirect Costs | Site & Sponsor Staff Retraining | (Labor Hours) | Diverts resources from ongoing trial activities; requires investigator meetings and protocol re-education [2]. |
| Project Management & Oversight | (Labor Hours) | Increased effort for change management, communication, and tracking compliance [2]. | |
| Hidden Costs | Timeline Extensions & Delays | $XXX,XXX | Implementation averages 260 days; lost opportunity cost and extended resource allocation [2]. |
| Patient Re-consent & Screening | Delays enrollment | Stalls patient enrollment and site activity, creating ripple effects on recruitment goals [2]. | |
| Statistical & Reporting Re-work | (Labor Hours) | Impacts TLFs and statistical analysis plans; alters resource allocation and final deliverables [2]. |
Necessary vs. Avoidable Amendments
Not all amendments are created equal. Research suggests that 23% of amendments are potentially avoidable, meaning better protocol planning could save substantial time and money [2]. Distinguishing between necessary and avoidable changes is key to cost containment.
Necessary Amendments are typically driven by external factors or critical new information:
- Safety-Driven Changes: Implementation of new adverse event monitoring requirements based on emerging data [2].
- Regulatory-Required Adjustments: Compliance with updated guidance from the FDA or EMA [2].
- New Scientific Findings: Incorporation of new biomarkers or stratification strategies based on peer-reviewed evidence [2].
Avoidable Amendments often stem from internal planning deficiencies:
- Protocol Title Changes: Alters the study title, requiring updates to all regulatory filings and creating unnecessary administrative burden [2].
- Shifting Assessment Time Points: Triggers budget renegotiations, database updates, and system revalidation for minimal scientific gain [2].
- Minor Eligibility Criteria Adjustments: Leads to patient reconsent, IRB resubmission, and operational complexity without materially improving scientific validity [2].
Diagram 1: Decision Pathway for Classifying Protocol Amendments. This workflow helps researchers determine whether a proposed change is a necessary or avoidable amendment, guiding appropriate management strategies.
Experimental Protocols for Cost Assessment
Accurately quantifying the financial impact of trial modifications requires rigorous, standardized methodologies. The following protocols provide a framework for collecting and analyzing cost data.
Micro-Costing Methodology for Clinical Trial Operations
Micro-costing is a bottom-up approach that involves direct enumeration and costing of each resource consumed in a process. It is considered the most accurate and precise method for deriving intervention costs, forming a foundation for reliable economic evaluation [14].
Primary Steps for Micro-Costing Protocol Amendments:
- Define the Intervention and Perspective: Clearly delineate the amendment process and adopt the study perspective (e.g., sponsor, site, or societal) [14].
- Identify Resource Inputs: Catalog all resources consumed due to the amendment. This includes personnel time, materials, and overhead. The "Research Reagent Solutions" table in Section 5 provides key tools for this process.
- Quantify Resource Units: Measure the volume of each resource used (e.g., hours of labor, number of forms, software license days).
- Assign Unit Costs: Attach a monetary value to each unit of resource, using internal financial data or standardized national cost databases.
- Aggregate and Analyze: Sum all costs and perform sensitivity analyses to test the robustness of the estimates against variations in key assumptions [14].
Data Collection Tools for Micro-Costing:
- Standardized Comprehensive Templates: Structured forms to capture all cost categories systematically.
- Targeted Questionnaires: Surveys designed for specific stakeholders (e.g., project managers, data coordinators, site staff) to gather data on time and resource utilization.
- Activity Logs: Diaries or digital trackers for staff to record time spent on amendment-related activities in real-time.
- On-site Administrative Data Extraction: Harvesting data from existing financial, ERP, and clinical trial management systems [14].
Cost-Comparison Analysis Framework
For a higher-level comparison of the financial impact of different trial management approaches (e.g., pre-amendment planning vs. post-amendment response), a cost-comparison analysis is appropriate. This method is detailed in guidelines from organizations like the UK's National Institute for Health and Care Excellence (NICE) [15].
Key Features of the Framework:
- Time Horizon: The analysis period must be long enough to capture all materially important cost differences. For amendments, this should cover from initiation through full implementation and stabilization [15].
- Cost Inputs and Assumptions: The minimum analysis must include the acquisition costs of any new technologies or services required. It should also capture differences in resource use, such as changes in monitoring frequency, administration, and follow-up activities [15].
- Handling Uncertainty: Conduct sensitivity and scenario analyses to describe the impact of varying inputs that are subject to uncertainty, such as personnel cost rates or the duration of implementation delays [15].
Diagram 2: Micro-Costing Workflow for Protocol Amendments. This workflow outlines the five primary steps for conducting a detailed bottom-up cost analysis, supported by specific data collection tools.
Strategic Management of Amendment Costs
Proactive management strategies can significantly reduce the frequency of avoidable amendments and mitigate the financial impact of necessary ones.
Prevention Strategies for Avoidable Amendments
- Engage Key Stakeholders Early: Involve regulatory experts, operational staff, site investigators, and even patient advisors during the initial protocol design phase. Using patient advisory boards can refine protocols and reduce mid-trial changes [2].
- Implement Protocol Quality by Design: Conduct rigorous feasibility assessments and risk analyses before finalizing the protocol. Leverage data from completed trials to identify common pain points that lead to amendments.
- Utilize Protocol Authoring Tools: Employ standardized templates and structured content guidance to improve the clarity, consistency, and completeness of initial protocols.
Mitigation Strategies for Necessary Amendments
- Bundle Amendments Strategically: Group multiple changes into planned update cycles rather than implementing them serially. This streamlines regulatory submissions and reduces administrative burden. However, safety-related changes requiring immediate action should not be delayed for bundling [2].
- Establish Dedicated Amendment Teams: Create specialized, cross-functional teams to manage the amendment process. This ensures consistency, improves efficiency, and prevents disruptions to core trial activities [2].
- Implement Clear Communication Frameworks: Standardize training materials and document management to ensure smooth amendment adoption across all trial sites. Maintain trial momentum by keeping all stakeholders informed and aligned [2].
Table: Cost-Benefit Analysis of Amendment Management Strategies
| Management Strategy | Primary Application | Estimated Cost Saving Potential | Key Implementation Considerations |
|---|---|---|---|
| Early Stakeholder Engagement | Prevention | High (Reduces amendment frequency) | Requires upfront time investment; may lengthen protocol finalization. |
| Protocol Quality by Design | Prevention | High (Reduces amendment frequency) | Dependent on access to historical trial data and feasibility expertise. |
| Strategic Amendment Bundling | Mitigation | Medium (Reduces per-amendment costs) | Risk of delaying critical safety updates; requires careful judgment. |
| Dedicated Amendment Teams | Mitigation | Medium (Improves efficiency) | Initial resource allocation needed; benefits scale with trial portfolio size. |
| Investment in Training & Tech | Both | Variable (Long-term ROI) | Higher initial capital outlay; requires change management for adoption. |
Table: Key Research Reagent Solutions for Cost Analysis
| Tool / Resource | Category | Primary Function in Cost Analysis |
|---|---|---|
| Micro-Costing Data Collection Templates | Methodology Tool | Standardized forms for systematically capturing all resource inputs (labor, materials, overhead) consumed during an amendment process [14]. |
| Activity-Based Costing (ABC) Software | Technology | Allocates indirect and overhead costs to specific activities or projects (like an amendment) based on their actual consumption of resources, providing more accurate cost assignments [14]. |
| Clinical Trial Management System (CTMS) | Technology | Tracks protocol deviation rates, site performance metrics, and amendment implementation timelines—key data points for quantifying operational impacts [2]. |
| Electronic Data Capture (EDC) System | Technology | Manages protocol version control, data entry workflows, and required system modifications; provides data on reprogramming efforts and validation timelines post-amendment [2]. |
| Supplier Sourcing Databases | Procurement Tool | AI-driven databases help quickly identify and vet reliable vendors for critical amendment-related services (e.g., new central labs, imaging providers), reducing procurement delays [13]. |
A nuanced understanding of direct, indirect, and hidden expenses is indispensable for effective clinical trial management, particularly in the context of protocol amendments. The data clearly demonstrates that while necessary amendments are an inherent part of drug development, avoidable amendments represent a significant and manageable financial drain. By implementing structured cost-assessment methodologies like micro-costing, employing strategic prevention and mitigation tactics, and leveraging modern technology tools, research organizations can transform their approach to cost management. The goal is not to eliminate all changes, but to create a disciplined, data-driven environment where every expenditure—whether direct, indirect, or hidden—is recognized, accounted for, and optimized. This rigorous financial discipline is ultimately what enables the sustainable innovation required to bring new therapies to patients.
In contemporary clinical research, protocol amendments have evolved from occasional necessities to a pervasive operational challenge. A 2025 study reveals that 76% of Phase I-IV trials now require at least one amendment, a significant increase from 57% just a decade prior [2]. Each amendment triggers a cascade of operational disruptions, extending timelines by an average of 260 days for implementation and keeping sites operating under different protocol versions for approximately 215 days [2]. This analysis examines how amendments generate costly ripple effects across clinical trial operations, comparing the impacts of avoidable versus necessary changes within a comprehensive cost framework.
The financial implications are substantial. Individual amendments now cost between $141,000 to $535,000 in direct expenses alone, with the Tufts Center for the Study of Drug Development estimating that avoidable amendments consume approximately $2 billion annually across the industry [2] [1]. Beyond direct costs, amendments create hidden operational burdens including regulatory resubmissions, site retraining, database reconfiguration, and statistical plan revisions that collectively degrade trial efficiency and delay patient access to new therapies.
Quantitative Impact: The Data Behind Amendment Delays
Frequency and Cost Analysis
Table 1: Amendment Incidence and Financial Impact Across Trial Phases
| Trial Phase | Protocols Requiring Amendments | Average Amendments per Protocol | Direct Cost per Amendment |
|---|---|---|---|
| Phase I | 76% | 2.0 | $141,000 - $335,000 |
| Phase II | 76% | 2.7 | $141,000 - $453,932 |
| Phase III | 76% | 3.5 | $141,000 - $535,000 |
| Phase IV | 76% | 2.3 | $141,000 - $335,000 |
Recent benchmarking studies reveal that nearly all trial phases now experience similar rates of amendment adoption, though later-phase trials accumulate more amendments per protocol with significantly higher implementation costs [2]. The upward trend in amendment frequency correlates strongly with increasing protocol complexity, particularly in specialized domains like oncology where 90% of trials require at least one amendment [2].
Table 2: Distribution of Amendment Causes and Avoidability
| Amendment Cause | Frequency | Typically Avoidable? | Primary Impact Area |
|---|---|---|---|
| New Safety Information | 19.5% | No | Safety Monitoring |
| Regulatory Requests | 18.6% | No | Documentation |
| Study Strategy Changes | 18.4% | Sometimes | Protocol Design |
| Protocol Design Flaws | 11.3% | Yes | Eligibility Criteria |
| Recruitment Difficulties | 9.0% | Yes | Site Operations |
| Adding New Sites/Locations | 16.0% | Sometimes | Site Management |
Research indicates that 23-34% of amendments are potentially avoidable, primarily stemming from protocol design flaws, unfeasible eligibility criteria, and recruitment challenges that could be addressed through improved planning [2] [1] [16]. The most common amendment changes affect patient population descriptions and eligibility criteria (16%), safety assessment procedures (12%), and general protocol information (10%) [1].
Timeline Disruption Metrics
The operational ripple effects of amendments extend far beyond immediate financial costs. Implementation cycles—from problem identification to full site adoption—require a median of 65 days [1]. This timeline has expanded in recent years, with current data showing sites now require approximately 215 days to fully operationalize amendments across all locations, during which time sites operate under different protocol versions, creating significant compliance risks [2].
The distribution of amendment timing further compounds these disruptions. Approximately 40% of all amendments occur before the first patient receives first dose, with this percentage rising to 52% in Phase I studies where protocol complexity often reveals design flaws during early implementation [1]. This distribution pattern suggests significant opportunities for front-loaded improvements in protocol planning and feasibility assessment.
The Ripple Effect: How Amendments Disrupt Trial Operations
Operational Workflow Disruption
The implementation of protocol amendments triggers a cascade of interdependent operational activities that disrupt normal trial workflows. The diagram below illustrates how a single amendment creates ripple effects across multiple functional areas and timelines:
Amendment Ripple Effect Workflow
This workflow visualization demonstrates how a single amendment initiates parallel processes across regulatory, operational, and data management functions. The critical path typically flows through regulatory resubmission and IRB review, which requires 48 days on average for substantial amendments [16], during which sites cannot implement changes. This mandatory waiting period creates downstream compression of subsequent activities including staff retraining, patient reconsent, and system updates, often leading to compliance risks when different sites implement changes at different times.
Site-Level Disruption Patterns
Clinical trial sites bear the brunt of amendment implementation burdens. Each amendment requires sites to:
- Update protocol-specific documentation including investigator site files, pharmacy manuals, and standard operating procedures
- Retrain clinical staff on revised procedures, with training effectiveness varying significantly across sites
- Reconsent current patients under revised protocols, a process that typically results in 5-15% patient dropout [2]
- Renegotiate budgets and contracts to accommodate changed procedures, which consumes an average of 25-40 hours of site coordinator time per amendment [2]
These operational burdens disproportionately affect site resources because 58% of amendment implementation costs derive from increased investigative site fees and 24% from contract change orders with CROs and other vendors [1]. Sites additionally face the challenge of maintaining trial momentum while managing amendment implementation, particularly when amendments affect eligibility criteria that necessitate re-screening of potential participants.
Experimental Framework: Studying Amendment Impacts
Research Methodology for Amendment Impact Analysis
The quantitative data presented in this analysis derives from a mixed-methods research approach employed across multiple recent studies:
Study 1: Tufts CSDD Benchmarking (2025)
- Sample: Phase I-IV protocols from mid-sized and large pharmaceutical companies
- Data Collection: Comprehensive amendment tracking across 3,410 protocols
- Analysis Method: Quantitative analysis of amendment incidence, direct costs, and timeline impacts
- Cost Methodology: Detailed accounting of vendor change orders, site fee adjustments, and internal FTE costs [2]
Study 2: NHS UK Sponsored Research (2023)
- Design: Explanatory sequential mixed methods
- Sample: 242 approved amendments from 53 clinical research studies
- Data Collection: Content analysis of amendment forms followed by semi-structured interviews with 11 trial stakeholders
- Analysis Method: Inductive coding of amendment changes and reasons, thematically analyzed against interview data [16]
Study 3: Protocol Amendment Benchmark (2024)
- Sample: 3,596 amendments containing 19,345 protocol modifications
- Data Collection: Categorization of 6,855 specific changes classified by type and avoidability
- Analysis Method: Root cause analysis and avoidability assessment by sponsor companies [2]
These methodologies provide complementary perspectives on amendment impacts, with commercial sponsor data (Tufts CSDD) revealing financial and operational consequences, while non-commercial research (NHS) offers insights into process inefficiencies and root causes.
The Scientist's Toolkit: Amendment Management Solutions
Table 3: Essential Resources for Effective Amendment Management
| Tool/Solution | Primary Function | Implementation Benefit |
|---|---|---|
| Stakeholder Engagement Platforms | Facilitates cross-functional protocol review | Reduces design flaws by 23% [2] |
| Feasibility Assessment Tools | Predictive analytics for site performance | Identifies recruitment risks pre-activation |
| Protocol Template Systems | Standardized design with validated endpoints | Decreases inconsistencies and omissions |
| Amendment Bundling Frameworks | Groups multiple changes into coordinated updates | Reduces administrative burden by 35% [2] |
| Electronic Trial Master File (eTMF) | Centralized document management | Accelerates regulatory submission timelines |
| Site Communication Portals | Standardized training and document distribution | Ensures consistent amendment implementation |
Contemporary amendment management requires both technological solutions and strategic processes. Leading organizations implement dedicated amendment management teams that establish predefined decision frameworks for assessing amendment necessity, implementation timing, and bundling opportunities [2]. These teams employ structured content authoring and artificial intelligence tools, including large language models (LLMs), to streamline document modification and ensure consistency across multiple amended documents [17].
Functional service provider (FSP) partnerships have emerged as particularly valuable for managing amendment-related resourcing challenges, with two-thirds of drug developers now leveraging FSP or hybrid FSO arrangements to maintain operational continuity during amendment implementation [17]. These partnerships provide access to specialized amendment management expertise while converting fixed internal costs into flexible operational expenditures.
Comparative Analysis: Avoidable vs. Necessary Amendments
Strategic Framework for Amendment Classification
The operational and financial impacts of amendments vary significantly based on their underlying causes and implementation characteristics. The following comparative analysis distinguishes between avoidable and necessary amendments based on root causes, operational impacts, and mitigation strategies:
Table 4: Comparative Analysis of Amendment Types
| Parameter | Avoidable Amendments | Necessary Amendments |
|---|---|---|
| Root Causes | Protocol design flaws, unfeasible eligibility criteria, recruitment planning errors, administrative errors | New safety information, regulatory agency requests, new scientific findings, changes in standard of care |
| Proportion of Amendments | 23-34% [2] [1] | 66-77% [1] |
| Typical Implementation Cost | $141,000 - $453,932 [2] [1] | $141,000 - $535,000 [2] |
| Common Examples | Protocol title changes, minor eligibility adjustments, assessment schedule modifications | New adverse event monitoring requirements, compliance with updated FDA/EMA guidance, biomarker-driven stratification [2] |
| Primary Impact Area | Study startup and early enrollment | Ongoing trial conduct and long-term follow-up |
| Mitigation Strategies | Enhanced protocol review, stakeholder engagement, feasibility assessment, template standardization | Risk management planning, safety surveillance, regulatory intelligence, adaptive design elements |
This comparative framework reveals that while necessary amendments typically address legitimate scientific and safety concerns, avoidable amendments predominantly stem from correctable process deficiencies in protocol development and feasibility assessment. The data indicates that avoidable amendments cluster disproportionately in early trial phases and during study startup periods, with 52% of Phase I amendments occurring before first patient first dose compared to 30% of Phase III amendments [1].
Cost-Benefit Analysis of Amendment Prevention
Investments in amendment prevention demonstrate compelling returns based on multiple studies:
- Stakeholder Engagement Return: Protocols developed with comprehensive cross-functional input experience 2.0 average amendments compared to 3.2 amendments for more complex protocols developed without adequate stakeholder engagement [1]
- Feasibility Assessment Impact: Sites participating in early feasibility assessment demonstrate 28% faster amendment implementation and 15% higher protocol compliance following amendments [16]
- Protocol Template ROI: Standardized protocol templates with validated endpoints reduce amendment incidence by 18% primarily through reduced eligibility criterion modifications and assessment schedule changes [2]
The cumulative financial impact of amendment prevention is substantial. For a typical mid-sized pharmaceutical company managing 40-60 active protocols simultaneously, reducing avoidable amendments by 50% would yield approximately $12-18 million in annual savings based on current amendment cost benchmarks [2] [1]. These savings primarily derive from reduced site fee increases, vendor change orders, and internal resource allocation.
Protocol amendments generate significant and predictable ripple effects across clinical trial operations, with documented impacts on timelines, costs, and site efficiency. The distinction between avoidable and necessary amendments provides a strategic framework for targeted process improvements, with approximately one-third of amendments presenting prevention opportunities through enhanced protocol planning and feasibility assessment [2] [1].
Successful amendment management requires both preventive strategies—including comprehensive stakeholder engagement and protocol review—and implementation efficiencies through dedicated amendment teams and strategic bundling approaches. Organizations that systematically address amendment prevention and optimization can realize substantial operational and financial benefits while accelerating clinical development timelines and reducing site burden.
As clinical trial complexity continues to increase, particularly in specialized therapeutic areas like oncology and rare diseases, proactive amendment management will become increasingly critical to maintaining development efficiency and ensuring timely patient access to innovative therapies.
A Tactical Guide to Identifying and Categorizing Amendment Triggers
Clinical trial protocol amendments are a pervasive and costly challenge in drug development. Recent research indicates that 76% of Phase I-IV trials require at least one amendment, a significant increase from 57% in 2015 [2]. Among these, a substantial portion—estimated between 23% and 45%—are considered avoidable, stemming from correctable issues in protocol design and planning [2] [5]. This analysis compares the financial and operational impact of avoidable amendments against necessary ones, demonstrating that strategic investments in protocol quality and feasibility assessment can prevent significant budget overruns and timeline extensions, thereby optimizing drug development efficiency.
A protocol amendment is a formal change to the clinical trial protocol after it has received initial approval from an Institutional Review Board (IRB) or regulatory authority. Amendments are categorized as either "substantial" (global changes requiring re-approval) or "non-substantial" [18]. While some amendments are unavoidable responses to new scientific data or regulatory requirements, a critical distinction exists between those that are necessary and those that are preventable through improved processes.
The high frequency of amendments signals systemic issues in protocol development. Phase III trials average 2.3 amendments per protocol, with direct implementation costs reaching a median of $535,000 each [2] [19]. The downstream operational impacts, including enrollment freezes and timeline extensions, create even greater financial burdens and delays in bringing new treatments to patients.
Quantitative Impact: Avoidable vs. Necessary Amendments
Prevalence and Direct Costs
Table 1: Amendment Prevalence and Direct Costs by Trial Phase
| Trial Phase | Protocols with ≥1 Amendment | Mean Amendments per Protocol | Median Direct Cost per Amendment |
|---|---|---|---|
| Phase I - IIIb/IV | 57% [18] | 2.1 [18] | Not Specified |
| Phase II | 77% [18] | 2.2 [18] | $141,000 [18] |
| Phase III | 66% [18] | 2.3 [18] | $535,000 [19] |
| Phase I-IV (2024) | 76% [2] | Not Specified | $141,000 - $535,000 [2] |
Table 2: Classification and Avoidability of Substantial Amendments
| Amendment Category | Proportion | Common Causes |
|---|---|---|
| Completely Avoidable | 23% [18] | Protocol design flaws, narrative errors, infeasible eligibility criteria [18] |
| Somewhat Avoidable | 22% [18] | Infeasible execution instructions, recruitment challenges [18] |
| Somewhat Unavoidable | 30% [18] | Manufacturing changes, new safety data [18] |
| Completely Unavoidable | 25% [18] | Regulatory agency requests, changes in standard of care [18] |
Operational and Timeline Impact
The indirect costs of amendments often surpass direct expenses. Protocols with at least one substantial amendment experience significantly longer cycle times [18]:
- → Protocol approval to Last Patient Last Visit (LPLV): 90 days longer
- → First Patient First Visit (FPFV) to LPLV: 85 days longer
- → First Patient Participation Cycle (FPFV to FPLV): ~5.5 months (165 days) longer
Amendments also severely impact patient enrollment. Amended protocols screened 291 fewer patients than planned on average, compared to just 57 fewer for protocols without amendments. Furthermore, amended trials enrolled 107 fewer patients than planned, while unamended trials actually enrolled 49 more than planned [19].
Root Causes of Avoidable Amendments
The following diagram illustrates the primary pathways leading to avoidable amendments, originating from foundational flaws in protocol development.
Infeasible Eligibility Criteria (53% of amendments)
Root Cause: Overly restrictive or scientifically unjustified patient selection criteria that do not reflect real-world patient populations or healthcare practices [2] [5].
Experimental Protocol for Validation: To prevent this, sponsors conduct eligibility criteria feasibility analysis using:
- Retrospective Data Analysis: Analyzing de-identified electronic health records (EHR) from target regions to quantify the number of patients meeting all proposed criteria.
- Site Feasibility Surveys: Surveying principal investigators at potential trial sites about the feasibility of finding eligible patients based on their clinical experience.
- Standard of Care (SoC) Alignment Checks: Comparing proposed criteria against local treatment pathways to ensure required prior therapies, washout periods, and biomarker testing are consistent with real-world practice [5].
Flaws in Trial Design and Procedures (38% of amendments)
Root Cause: Unworkable visit schedules, complex assessment procedures, or misalignment between endpoints and corresponding procedures that sites cannot execute or patients cannot comply with [2] [5].
Experimental Protocol for Validation: Leading organizations implement protocol simulation and burden assessment:
- Patient Advisory Boards: Convening 6-10 patients from advocacy groups to review and simulate the proposed schedule of assessments, providing feedback on burden and feasibility [5] [18].
- Site Simulation Exercises: Having clinical research coordinators walk through every procedure in the protocol to identify logistical conflicts, excessive burden, or need for specialized equipment.
- Comparator Drug SoC Analysis: Verifying that the chosen comparator drug is commercially available, reimbursed, and part of local treatment guidelines in all target countries [5].
Protocol Inconsistencies and Errors (35% of amendments)
Root Cause: Typographical errors, contradictory instructions between the protocol narrative and flow charts, or misalignment between endpoints and statistical analysis plans [18].
Experimental Protocol for Validation: Implementing systematic protocol quality control:
- Cross-Functional Review Teams: Establishing mandatory review cycles with representatives from clinical operations, biostatistics, data management, regulatory affairs, and patient engagement.
- Common Protocol Template Adoption: Using standardized templates (e.g., TransCelerate Common Protocol Template) to ensure consistent structure and language, making misalignments easier to identify [18].
- Governance Committee Review: Requiring senior-level governance committee sign-off to ensure protocol consistency with the overall development plan and challenge executional feasibility [18].
The Researcher's Toolkit: Solutions for Amendment Prevention
Table 3: Essential Resources and Methodologies for Protocol Optimization
| Tool / Methodology | Function | Implementation Example |
|---|---|---|
| Stakeholder Engagement Platforms | Facilitates early input from patients, site staff, and advocacy groups to identify feasibility issues. | Eli Lilly conducts patient simulation exercises to observe participation experience and address burdens before protocol finalization [18]. |
| Standard of Care (SoC) Databases | Provides data on local treatment pathways, reimbursement status, and drug availability to align protocol with real-world practice. | Automated SoC insights pulled from local reimbursement authorities prevent amendments related to comparator drug feasibility [5]. |
| Protocol Review Committees | Formal governance mechanism for cross-functional expert review to ensure consistency and feasibility. | Pfizer's revised SOP requires detailed protocol review before implementation; Amgen uses a Development Design Center for expert consultation [18]. |
| Feasibility Assessment Tools | Quantifies patient population availability and site capability to execute protocol requirements. | Retrospective EHR analysis and site feasibility surveys quantify eligible patient numbers and identify procedural barriers. |
| Common Protocol Templates | Standardizes protocol structure and language to reduce errors and inconsistencies. | TransCelerate BioPharma's Common Protocol Template helps identify misalignment between endpoints and procedures [18]. |
The data presents a compelling case for re-engineering protocol development processes. With nearly half of all substantial amendments being preventable, the industry has a significant opportunity to improve clinical trial efficiency. The cost differential is stark: investing in robust protocol design through stakeholder engagement, feasibility analysis, and quality control processes yields substantial returns by avoiding $500,000+ amendment costs and 3-6 month timeline extensions [2] [18].
Organizations that master this balance stand to gain significant advantages through improved trial performance and reduced operational costs. As development pipelines grow increasingly complex, particularly in oncology and rare diseases where amendment rates reach 90%, building prevention-oriented approaches becomes not merely an efficiency initiative but a strategic imperative for sustainable drug development [2].
In contemporary clinical research, a well-defined patient pathway is a critical determinant of trial success. The design of these pathways, which outline the sequence of care and data collection points, has direct and substantial financial implications. Poorly planned pathways frequently lead to protocol amendments, with a recent study revealing that 76% of Phase I-IV trials require at least one amendment, a significant increase from 57% in 2015 [2]. Each amendment carries a staggering price tag, costing sponsors between $141,000 and $535,000 in direct expenses, not accounting for indirect costs from delayed timelines and operational disruptions [2]. This article compares traditional, opinion-based pathway design against emerging, data-driven methodologies, framing the analysis within the critical context of avoiding costly, avoidable amendments while facilitating necessary ones.
Comparative Analysis: Traditional vs. Data-Driven Pathway Design
The following table objectively compares the core characteristics of the two dominant approaches to patient pathway design.
Table 1: Comparison of Pathway Design Methodologies
| Feature | Traditional Clinical Pathways (CPWs) | Data-Driven Clinical Evidence-Based Pathways (CEBPWs) |
|---|---|---|
| Basis of Design | Practical experience and consensus of clinical experts [20]. | Objective analysis of real-world, patient-level data from hospital systems [20]. |
| Data Foundation | Limited, often subjective; manual curation is slow and costly [20]. | Large-scale clinical datasets; the TuPro study generated up to 500 Gb of data per sample [21]. |
| Quantitative Performance | Lower case coverage; generates variability and implementation bias [20]. | Achieves 100% coverage in diagnostics and therapy; 81.81% drug coverage [20]. |
| Flexibility & Personalization | Rigid, standardized treatment modes; poorly adapted to individual patient needs [20]. | Accommodates individual patient characteristics while reflecting population-level data trends [20]. |
| Impact on Amendments | Higher potential for avoidable amendments due to design flaws discovered mid-trial [2]. | Aims to reduce avoidable amendments by creating more robust, evidence-based initial protocols [2] [20]. |
| Primary Utility | Defining a general standard of care. | Informing treatment decisions, detecting irregularities, and supporting insurance payment standards [20]. |
Quantitative Impact: The Amendment Cost Framework
Understanding the financial impact of protocol changes is essential for evaluating the return on investment of improved pathway design. The table below breaks down the costs associated with amendments, which can be mitigated by more feasible initial designs.
Table 2: Financial and Operational Impact of Protocol Amendments
| Cost Category | Quantitative Impact | Examples of Triggering Events |
|---|---|---|
| Direct Amendment Cost | $141,000 - $535,000 per amendment [2]. | Safety concerns, new regulatory requirements, new scientific findings [2]. |
| Timeline Impact | Implementation now averages 260 days; sites operate under different protocol versions for 215 days on average [2]. | Changing assessment timepoints; minor eligibility criteria adjustments [2]. |
| Operational & Regulatory Costs | IRB resubmission fees and weeks of delay; site budget renegotiations; data management system updates [2]. | Protocol title changes; shifting a single assessment schedule; minor eligibility adjustments [2]. |
| Avoidable Amendments | 23% of amendments are potentially avoidable through better protocol planning [2]. | Poor initial protocol design, rushed decision-making, failure to engage key stakeholders early [2]. |
Experimental Protocol: The CEBPW Development Workflow
The development of Clinical Evidence-Based Pathways (CEBPWs) represents a foundational data-driven methodology. The following workflow is derived from a large-scale study that utilized hospital data from 166 public hospitals in Shanghai, involving 5.3 million cases [20].
- Data Acquisition and Curation: Collect hospitalised patient-level data, including front-sheet medical records, detailed hospitalisation charges, and outpatient charges. All personal identifiers are removed [20].
- Data Preprocessing and Dimension Reduction: Combine databases using unique identification numbers. Categorize and label all items as diagnosis, therapy, drugs, or consumables. Reduce the dimensionality of the original charge data while retaining main features [20].
- Frequent Pattern Mining: Apply the FP-GROWTH algorithm (using the
arulespackageeclatfunction with a support parameter of 0.7) to identify combinations of frequently charged items within each 'diagnosis+therapy' group. This step identifies:- Mandatory Items: Procedures for which there is 100% patient adherence.
- High-Frequency Items: Procedures for which there is 95% patient adherence.
- Low-Frequency Items: Procedures for which there is 30% patient adherence, determined by individual patient characteristics [20].
- Pathway Generation and Clustering: The FP-GROWTH algorithm generates multiple potential pathways. The distribution probability of all data is calculated, and patient data is converted into a normal distribution. Gaussian clustering is used for data merging, and a time dimension is superimposed to sequence activities [20].
- Similarity Calculation and Calibration: Use cosine similarity and Pearson correlation coefficients to study the conformity and fit between each generated pathway and the real patient data, thereby calibrating the model [20].
Diagram 1: CEBPW Development Workflow
Advanced Application: Multiomics Profiling for Precision Pathways
For complex diseases like cancer, patient pathways are moving beyond standardized care into highly personalized regimens. The prospective Tumor Profiler (TuPro) study on melanoma demonstrates a cutting-edge methodology for guiding these precision pathways.
Experimental Protocol: Multiomics Tumor Profiling
The TuPro study employed a multi-technology, single-cell analysis pipeline to inform treatment decisions [21].
- Patient Cohort and Biopsy: Enroll eligible patients (e.g., n=116 for melanoma). Obtain tumor biopsies (e.g., 126 samples including longitudinal) under ethical approval [21].
- Parallel Multiomics Analysis: Process samples using multiple independent technologies within a strict turnaround time (e.g., 4 weeks). Core technologies include:
- Single-cell Genomics (scDNA-seq) & Transcriptomics (scRNA-seq)
- Targeted Proteomics: Imaging Mass Cytometry (IMC) and Cytometry by Time of Flight (CyTOF)
- Proteotyping: using Data-Independent Acquisition (DIA)
- Drug Phenotyping: Pharmacoscopy and iterative indirect immunofluorescence imaging (4iDRP)
- Targeted Next-Generation DNA Sequencing (NGS)
- Digital Pathology (DigiPath) [21]
- Data Integration and Molecular Tumor Board (MTB) Review: Integrate the multiomics data (up to 40,000 potential markers per sample) with clinical and diagnostic information. A multidisciplinary MTB reviews the data and selects relevant biomarkers (e.g., 54 markers in the TuPro study) to inform treatment recommendations [21].
- Treatment Decision and Outcome Tracking: Patients receive either Standard of Care (SOC) treatments or highly individualized, poly-biomarker-driven treatments beyond SOC. Objective response rate, disease control rate, and progression-free survival are tracked [21].
Diagram 2: Multiomics Profiling for Therapy
Performance Data from the TuPro Study
The TuPro study provides quantitative evidence of the feasibility and utility of this data-driven approach [21]:
- Feasibility: The complex analytical workflow was completed within a 4-week turnaround time from biopsy to MTB reporting.
- Utility: In 75% of cases, the multiomics data were judged useful for informing treatment recommendations.
- Efficacy: For patients receiving beyond-SOC treatment (n=37), the objective response rate was 38%, with a disease control rate of 54%.
- Survival: The median progression-free survival for patients with TuPro-informed therapy was 6.04 months, even in later therapy lines [21].
The Scientist's Toolkit: Essential Reagents & Technologies
Table 3: Key Research Reagent Solutions for Data-Driven Pathway Design
| Reagent / Technology | Function in Pathway Design & Validation |
|---|---|
| FP-GROWTH Algorithm | An efficient data mining algorithm for finding frequent patterns (e.g., common diagnostic-therapy combinations) in large clinical datasets [20]. |
| Imaging Mass Cytometry (IMC) | A targeted proteomics technology that allows for multiplexed imaging of tumor tissues at single-cell resolution, revealing cellular heterogeneity and spatial relationships [21]. |
| Pharmacoscopy | A high-content imaging method that tests drug susceptibility directly on primary patient tumor cells, providing functional drug phenotyping data [21]. |
| Single-cell RNA Sequencing (scRNA-seq) | Provides a transcriptomic profile of individual cells within a tumor, identifying distinct cell populations and their functional states [21]. |
| Activity Tracker (Connected Watch) | In behavioral or quality-of-life studies, this tool objectively quantifies physical activity levels (e.g., step counts) to correlate with patient outcomes and refine supportive care pathways [22]. |
The evidence demonstrates a clear paradigm shift in clinical trial design. Traditional, opinion-based patient pathways are inherently prone to the costly amendments that impact 76% of trials. In contrast, methodologies like Clinical Evidence-Based Pathways (CEBPWs) and multiomics tumor profiling leverage real-world and deep molecular data to create more robust, feasible, and personalized patient pathways. The initial investment in these data-driven approaches offers a substantial return by minimizing avoidable amendments—which constitute 23% of all changes—and their associated costs, which can reach half a million dollars each. For researchers and drug development professionals, the strategic integration of SoC and novel data sources is no longer optional but essential for designing efficient trials, controlling development costs, and ultimately, delivering better therapies to patients.
In the world of clinical development, protocol amendments represent a significant and growing challenge to operational efficiency and financial performance. Recent data indicate that 76% of Phase I-IV trials now require amendments, a substantial increase from 57% in 2015 [2]. These changes carry staggering price tags, with a single amendment costing between $141,000 and $535,000 in direct expenses alone, not including indirect costs from delayed timelines and operational disruptions [2].
This case study examines how one Top 10 pharmaceutical company nearly missed critical country-specific reimbursement data that could have triggered a catastrophic protocol amendment—and how a more sophisticated data verification approach helped them avert a potential $1.6 million loss. We frame this specific incident within the broader context of avoidable versus necessary amendments, providing drug development professionals with actionable insights for distinguishing between essential protocol changes and preventable operational missteps.
The Close Call: Averted Disaster in Rare Disease Trial Planning
The Scenario: Country Selection for a Rare Disease Trial
A Top 10 pharmaceutical company was planning a clinical trial for a rare disease indication, with initial country selection targeting eight countries [23]. Before finalizing their strategy, the team needed clarity on a critical factor: the reimbursement status of a monoclonal antibody that was already part of standard treatment options in these target countries [23]. This information was essential for protocol design, as it would directly impact patient recruitment, site selection, and standard of care assumptions.
The team ran a comparative analysis using two different data sources:
- Their legacy platform, which relied primarily on Health Technology Assessment (HTA) recommendations
- The TrialHub platform, which sources real-time data from National Competent Authorities (NCAs) that provide definitive reimbursement status [23]
The Discovery: Divergent Data Reveals Critical Flaw
The comparison revealed dramatically different pictures of the treatment landscape. The legacy platform indicated the drug was reimbursed in most target countries, while TrialHub's NCA-sourced data showed it was not actually reimbursed in several countries [23].
This discrepancy had profound implications. Had the team relied on their original data source, they would have launched the trial in countries where:
- Patient recruitment would have been severely impacted
- Standard of care assumptions in the protocol would have been incorrect
- A substantial protocol amendment would have been inevitable to address these issues [23]
The Financial Impact: Calculating the Averted Costs
The near-mistake would have necessitated at least one substantial protocol amendment. Given that the trial was progressing toward Phase III, the potential financial impact was significant [23]:
| Trial Phase | Cost per Amendment | Mean Number of Amendments | Potential Total Cost |
|---|---|---|---|
| Phase II | $141,000+ | 2.2 | $310,200+ |
| Phase III | $535,000+ | 2.3 | $1,230,500+ |
Table 1: Direct costs of protocol amendments by phase [23]
With combined Phase II and III costs, the total potential financial impact reached approximately $1.6 million in direct costs alone [23]. This calculation does not include indirect costs such as:
- Timeline delays extending to 260 days for amendment implementation [2]
- Recruitment challenges and associated delays
- Stakeholder pressure and reputational damage
- Poor patient experience and frustrated sites [23]
Understanding Protocol Amendments: A Necessary Nuisance
The Amendment Landscape: Incidence and Causes
Protocol amendments are changes made to a clinical trial after it has received regulatory approval [6]. Recent research shows that nearly 60% of protocols require one or more amendments, with later-phase protocols averaging 2.7 (Phase II) to 3.5 (Phase III) amendments each [1].
The causes of amendments fall into two broad categories:
Necessary Amendments (Approximately 66%)
- New safety information (19.5%)
- Regulatory agency requests (18.6%)
- Changes in standard of care
- New scientific findings [1] [2]
Avoidable Amendments (Approximately 34-45%)
- Protocol design flaws and inconsistencies (11.3%)
- Difficulties recruiting study volunteers (9%)
- Lack of understanding of patient standard of care
- Rushed initial applications [1] [6] [23]
The True Cost of Amendments: Beyond Direct Expenses
While direct implementation costs are substantial, the full impact of amendments extends far beyond budgetary considerations:
| Cost Category | Specific Impacts |
|---|---|
| Direct Costs | - IRB review fees- CRO contract change orders (24% of costs)- Investigative site fee increases (58% of costs)- Regulatory submission fees |
| Operational Costs | - Staff retraining and protocol re-education- Data management and system updates- Statistical analysis plan revisions- Document translation and updates |
| Timeline Impacts | - Median 65-day cycle time per amendment- 260-day implementation timeline- Sites operating under different protocol versions for 215 days |
| Strategic Costs | - Delayed commercialization- Competitive disadvantages- Strain on stakeholder relationships |
Table 2: Comprehensive costs of protocol amendments [2] [1]
The Tufts Center for the Study of Drug Development estimates that the total cost for sponsors to implement "avoidable" protocol amendments each year is approximately $2 billion in direct costs alone, not including billions in potential time-savings from earlier commercialization [1].
Experimental Protocols: Methodologies for Amendment Analysis
Amendment Root Cause Analysis Methodology
Understanding what drives amendments requires systematic analysis. The following methodology was adapted from established research approaches [6]:
Data Collection Protocol
- Collect amendment forms and supporting documentation for a representative sample of trials
- Extract individual amendment changes and reasons where stated
- Code changes and reasons using conventional content analysis approaches
- Group codes into content-related categories
- Perform statistical analysis to identify frequency patterns
Stakeholder Interview Protocol
- Conduct semi-structured interviews with trial stakeholders
- Explore views on reasons underpinning amendment submission
- Identify potential efficiencies that could prevent avoidable amendments
- Analyze transcripts using thematic analysis with a framework approach
This mixed-methods approach allows researchers to quantify amendment patterns while gaining qualitative insights into their root causes.
Feasibility Assessment Methodology
The case study highlights the critical importance of accurate feasibility assessment. The following workflow illustrates the decision process that prevented the $1.6M amendment:
Diagram 1: Feasibility assessment workflow
This systematic approach to data verification enabled the pharmaceutical company to identify inaccurate assumptions before protocol finalization, preventing a costly mid-trial correction.
Comparative Analysis: Avoidable vs. Necessary Amendments
Distinguishing between necessary and avoidable amendments is crucial for effective trial management. The table below compares their key characteristics:
| Characteristic | Necessary Amendments | Avoidable Amendments |
|---|---|---|
| Primary Causes | - New safety information- Regulatory requirements- New scientific findings- Changes in standard of care | - Protocol design flaws- Infeasible eligibility criteria- Lack of understanding of local standard of care- Rushed initial planning |
| Typical Timing | Distributed throughout trial timeline | Often occur before first patient first dose (30-52% across phases) |
| Financial Impact | Justified investment for trial integrity/success | Pure cost with minimal scientific value |
| Prevention Strategy | Difficult to prevent; focus on efficient implementation | Highly preventable through better planning and feasibility assessment |
| Examples | - New adverse event monitoring- Compliance with updated FDA guidance- Biomarker-driven stratification | - Changing protocol titles- Shifting assessment time points- Minor eligibility criteria adjustments- Incorrect standard of care assumptions |
Table 3: Characteristics of necessary vs. avoidable amendments [2] [1] [6]
The Scientist's Toolkit: Research Reagent Solutions for Protocol Optimization
Drug development professionals can leverage several tools and methodologies to minimize avoidable amendments:
| Tool/Solution | Function in Amendment Prevention |
|---|---|
| Real-Time Reimbursement Data Platforms | Provide accurate, country-specific standard of care and reimbursement data to inform feasible protocol design |
| Artificial Intelligence (AI) in Protocol Design | Speed identification of drug candidates and predict potential design flaws through in-silico modeling |
| Structured Feasibility Assessment Frameworks | Systematically evaluate protocol feasibility before finalization through stakeholder engagement and data verification |
| Patient Advisory Boards | Incorporate patient perspectives to refine protocols and reduce mid-trial changes related to recruitment or burden |
| Next-Generation Sequencing (NGS) | Efficiently screen patient populations for trial eligibility, reducing need for eligibility criteria amendments |
| Digital Feasibility Platforms | Automate standard of care assessment across multiple countries to prevent site-level implementation issues |
Table 4: Research reagent solutions for protocol optimization [24] [23]
Strategic Framework for Amendment Management
Prevention Strategies: Proactive Protocol Planning
Based on the case study and supporting research, organizations can implement several key strategies to minimize avoidable amendments:
Engage Key Stakeholders Early
- Involve regulatory experts, site staff, and patient advisors at protocol development stage
- Use patient advisory boards to refine protocols and reduce mid-trial changes [2]
- Conduct critical reviews by various stakeholders with sufficient time allocated to planning [6]
Implement Robust Feasibility Assessment
- Move beyond HTA data to verify actual standard of care through National Competent Authorities
- Assess country-specific reimbursement landscapes before protocol finalization [23]
- Allocate sufficient time for planning and feasibility assessments rather than rushing applications [6]
Simplify Protocol Design
- Reduce protocol complexity, which correlates strongly with amendment incidence [1]
- Critically evaluate necessity of each procedure and eligibility criterion
- Balance scientific ambition with operational practicality
Management Strategies: Efficient Amendment Implementation
When amendments are necessary, structured implementation can minimize disruption:
Establish Dedicated Amendment Teams
- Assign specialized teams to manage amendment processes for consistency
- Prevent disruptions to ongoing trial activities by maintaining a structured approach [2]
Bundle Amendments Strategically
- Group multiple changes into planned update cycles to streamline regulatory submissions
- Balance efficiency with regulatory compliance, especially for safety-driven changes [2]
Implement Clear Communication Frameworks
- Standardize training and document management to ensure smooth amendment adoption
- Maintain trial momentum by keeping all stakeholders informed and aligned [2]
The case of the Top 10 pharmaceutical company's near-$1.6 million mistake illustrates the critical importance of accurate data and thorough feasibility assessment in clinical development. While some amendments will always be necessary to respond to new scientific information and ensure patient safety, a significant proportion—estimated at 34-45%—can be prevented through better planning, stakeholder engagement, and verification of key assumptions [1] [23].
As clinical trials grow increasingly complex, particularly in oncology and rare diseases where 90% of trials require at least one amendment, the ability to distinguish between necessary and avoidable changes becomes increasingly critical [2]. By implementing the frameworks and tools outlined in this analysis, drug development professionals can enhance trial efficiency, reduce financial waste, and ultimately bring new treatments to patients faster.
The $1.6 million savings in this case study represents more than just financial preservation—it demonstrates how strategic investment in proper planning and verification creates value across the clinical development lifecycle, from sponsor to site to patient.
This guide provides an objective comparison of the outcomes associated with rigorous versus underdeveloped feasibility and protocol review processes. The analysis is framed within a broader thesis on the cost comparison of avoidable versus necessary amendments in clinical research, utilizing available data on development costs and feasibility study frameworks.
Quantitative Context: Drug Development Costs and Feasibility
The high cost of drug development underscores the critical importance of robust feasibility assessment to avoid wasteful expenditures. The following table summarizes key financial metrics.
Table 1: Selected Pharmaceutical Research and Development (R&D) Cost Metrics
| Metric | Reported Value | Context and Source |
|---|---|---|
| Average R&D Cost per Asset (2024) | $2.23 billion [25] | Based on analysis of 20 large pharma companies; costs increased from $2.12B in 2023 [25]. |
| Median Direct R&D Cost per New Drug | $150 million [26] | RAND study of 38 FDA-approved drugs; mean cost was $369M, indicating skew by high-cost outliers [26]. |
| Cost of Terminated Candidates (2024) | $7.7 billion [25] | Amount spent by a cohort of 20 pharmas on trials for candidates that were terminated [25]. |
| Forecast Average Internal Rate of Return (IRR) | 5.9% (2024) [27] | Improvement from 4.1% in 2023; excludes GLP-1 therapies, the average drops to 3.8% [25]. |
Comparative Checklists: Rigorous vs. Inadequate Review Processes
A thorough pre-submission review is a necessary investment to prevent avoidable amendments. The table below contrasts characteristics of rigorous and inadequate approaches.
Table 2: Checklist for Pre-Submission Feasibility and Protocol Review
| Review Domain | Rigorous Review Process (Mitigates Avoidable Costs) | Inadequate Review Process (Risks Avoidable Costs) |
|---|---|---|
| Protocol Design Feasibility | Protocol aligns with local standard of care; inclusion/exclusion criteria are workable; patient burden is minimized [28]. | Complex protocol is incompatible with local practices; overly restrictive criteria hinder enrollment [28]. |
| Patient Recruitment Feasibility | Realistic assessment of prevalence and recruitment potential using historical data and electronic records; diverse strategies [28]. | Overestimation of eligible patients; reliance on subjective site estimates; poor screening strategy [28]. |
| Operational & Site Feasibility | Site capabilities (staff, equipment) and track record with relevant trials are validated; complex protocol management is assessed [28]. | Site infrastructure and expertise are assumed or not fully evaluated; lack of specialized equipment [28]. |
| Regulatory & Ethical Feasibility | Local regulatory approval processes and timelines are reviewed; study aligns with ethical guidelines [28]. | Regulatory complexities are overlooked; potential for ethical challenges during conduct [28]. |
| Defined Aims & Objectives | Clear feasibility objectives are stated (e.g., recruitment rate, acceptability) to inform main trial design [29] [30]. | Aims focus on clinical effectiveness, failing to address key uncertainties for the main trial [29]. |
Experimental Protocols for Feasibility Assessment
Implementing a structured methodology for feasibility assessment is key to generating actionable data.
Protocol for a Comprehensive Clinical Trial Feasibility Study
This protocol outlines a multi-level assessment process to inform go/no-go decisions and protocol optimization [28].
1. Pre-Assessment Planning
- Define Objectives: Establish clear, quantitative objectives (e.g., "Determine if 300 patients can be recruited across 15 sites in 9 months") [28].
- Identify Stakeholders: Convene an interdisciplinary team with expertise in clinical operations, medical affairs, regulatory affairs, and data analytics [28].
- Select Sites: Compile a list of potential sites based on performance history, location, and therapeutic expertise [28].
2. Data Collection and Evaluation
- Epidemiology and Recruitment: Collect data on disease prevalence and patient demographics. Analyze historical recruitment data from similar studies, including screening failure rates [28].
- Site Capabilities: Assess infrastructure, staff expertise, and resources. For complex trials (e.g., involving biologics), verify capabilities like cold chain management [28].
- Regulatory Landscape: Review country-specific regulatory approval processes, timelines, and ethical guidelines [28].
- Protocol Alignment: Evaluate if the protocol is acceptable and compatible with local clinical practices and patient journeys [28].
3. Reporting and Decision-Making
- Compile and Interpret Data: Organize data into a coherent format using spreadsheets or visualization tools. Identify key challenges and opportunities [28].
- Develop Recommendations: Produce a comprehensive report with a SWOT analysis or similar output. Recommendations may include protocol modification, site selection, or expansion into new regions [28].
Protocol for a Pilot Implementation Trial
Pilot trials test implementation strategies on a smaller scale and can use specific hybrid designs [30] [31].
- Hybrid Type 1 Design: The primary aim is to test the effectiveness of a clinical/intervention on patient outcomes. The secondary aim is to gather preliminary information on the feasibility of implementing the intervention in a real-world setting [30].
- Hybrid Type 2 Design: Co-primary aims simultaneously test the clinical intervention and the feasibility or preliminary effectiveness of a structured implementation strategy [30].
Figure 1: Pathway for Selecting a Pilot or Feasibility Study Design.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key resources for designing and reporting high-quality feasibility and pilot studies.
Table 3: Essential Resources for Feasibility and Pilot Research
| Tool or Resource | Primary Function | Relevance to Feasibility/Protocol Review |
|---|---|---|
| CONSORT Extension for Pilot Trials [29] | Reporting Guideline | Provides a checklist to ensure transparent and complete reporting of pilot trial results, which should inform protocol development [29] [31]. |
| SPIRIT Guideline [29] | Protocol Reporting Standard | Defines items that should be included in a clinical trial protocol to ensure scientific and ethical rigor; can be adapted for pilot trials [29]. |
| Feasibility Assessment Questionnaire | Data Collection Tool | Customized, trial-specific instrument to gather objective data from potential sites on recruitment, capabilities, and protocol alignment [28]. |
| Electronic Medical Record (EMR) Data | Recruitment Estimation | Provides precise, data-driven estimates of eligible patient population size at a site, improving recruitment forecasting accuracy [28]. |
| Real-World Data (RWD) & Analytics | Evidence Generation | Informs trial design, patient selection, and site identification through analysis of real-world clinical data, supporting evidence-based decision-making [27] [25]. |
Figure 2: Logical relationship between assessment inputs, review quality, and amendment outcomes.
Proactive Prevention and Strategic Management of Amendments
In clinical development, protocol amendments are a major driver of escalating costs and timelines. Recent benchmarks reveal that over half of all clinical trials require at least one amendment, with some analyses indicating this figure has risen to 76% of Phase I-IV trials [32] [2]. The financial impact is substantial, with a single amendment costing between $141,000 and $535,000 in direct expenses, while complex Phase III trials can incur over $1.2 million per protocol change [2] [5]. Perhaps most strikingly, research indicates that 34-45% of amendments are avoidable, representing a significant opportunity for cost containment through improved planning and stakeholder engagement [32] [5].
This guide examines how early engagement of site staff and patient advocates can reduce avoidable amendments, comparing this preventive approach against the reactive costs of necessary amendments. By objectively analyzing experimental data and implementation methodologies, we provide a framework for quantifying the return on investment from strategic stakeholder integration in protocol design.
Quantitative Analysis: Amendment Costs and Frequency
Amendment Incidence and Financial Impact Across Trial Phases
Table 1: Protocol Amendment Frequency and Direct Costs by Trial Phase
| Trial Phase | Amendment Incidence | Average Amendments per Protocol | Direct Cost per Amendment | Total Direct Amendment Cost per Protocol |
|---|---|---|---|---|
| Phase I | Not specified | 2.0 (all phases) [1] | $141,000 (low estimate) [5] | $282,000 (estimated) |
| Phase II | 89% [4] | 2.2 [5] | $141,000 (median) [5] | $310,200 (median) [5] |
| Phase III | 75% [4] | 2.3 [5] | $535,000 (median) [5] | $1,230,500 (median) [5] |
| All Phases | 57-76% [32] [2] | 2.3-2.7 [32] [1] | $141,000-$535,000 [2] [5] | $453,932 (average) [1] |
Table 2: Breakdown of Amendment Implementation Costs by Activity Area
| Cost Category | Low Estimate | High Estimate | Percentage of Total Cost |
|---|---|---|---|
| IRB/EC Resubmission and Updates | $20,000 [32] | $50,000 [32] | 12.5-14.3% |
| Site/Vendor Retraining and Contracting | $50,000 [32] | $100,000 [32] | 31.3-28.6% |
| EDC Reprogramming | $25,000 [32] | $75,000 [32] | 15.6-21.4% |
| Subject Re-consenting and Coordination | $10,000 [32] | $25,000 [32] | 6.3-7.1% |
| Project Team Time (PMs, CRAs, etc.) | $50,000 [32] | $100,000 [32] | 31.3-28.6% |
| Total Estimated Cost | $160,000 [32] | $350,000 [32] | 100% |
Timeline Impact of Protocol Amendments
Beyond direct financial costs, amendments substantially prolong trial durations. Implementation of amendments now averages 260 days from internal approval to final ethics committee approval, with sites potentially operating under different protocol versions for over 215 days [32] [2]. Trials with at least one substantial amendment experience nearly three times longer durations from First Patient First Visit (FPFV) to Last Patient Last Visit (LPLV) compared to trials without such amendments [32].
Comparative Analysis: Avoidable vs. Necessary Amendments
Characteristics and Origins of Amendment Types
Table 3: Classification of Amendment Types and Their Drivers
| Amendment Characteristic | Avoidable Amendments | Necessary Amendments |
|---|---|---|
| Primary Drivers | Protocol design flaws [32], Inconsistencies in protocol [5], Unfeasible eligibility criteria [32] [5], Recruitment challenges [1] | New safety information [32] [1], Regulatory agency requests [32] [1], Changes in standard of care [1], New scientific findings [2] |
| Typical Examples | Changing protocol titles [2], Shifting assessment time points [2], Minor eligibility criteria adjustments [2], Addition of new trial sites [5] | Safety-driven changes (e.g., new AE monitoring) [2], Regulatory-required adjustments [2], Dose modifications based on new data [1] |
| Percentage of All Amendments | 34-45% [32] [5] | 55-66% [1] |
| Preventability | Preventable through better protocol planning and stakeholder engagement [32] [5] | Unavoidable, responding to external factors [1] |
Experimental Evidence: Stakeholder Impact on Amendment Reduction
Multiple studies have quantified the relationship between early stakeholder engagement and reduced amendment frequency:
Tufts CSDD Research: Studies found that protocols developed with multidisciplinary input during early development experienced significantly fewer amendments. The complexity-amendment correlation is clear: less complex protocols averaged two amendments, while more complex protocols averaged 3.2 amendments [1].
Oncology Trial Analysis: Research in specialized therapeutic areas demonstrated that nearly 90% of oncology trials require amendments, with many stemming from design issues that could be addressed through earlier site input on feasibility [2].
NHS-Sponsored Trials: An analysis of UK National Health Service trials found that 53% had at least one amendment, with a mean of 4.5 amendments per study. The single most cited reason was "to achieve the trial's recruitment target," suggesting inadequate initial assessment of patient enrollment feasibility [5].
Figure 1: Stakeholder Impact on Amendment Prevention - This workflow illustrates how early engagement influences amendment outcomes, showing that stakeholder input primarily reduces avoidable amendments while necessary amendments respond to external factors.
Methodological Framework: Stakeholder Engagement Protocols
Experimental Design for Stakeholder Integration
The following methodology outlines a structured approach for measuring the impact of stakeholder engagement on protocol feasibility:
Study Design: Prospective, controlled cohort study comparing amendment rates between protocols developed with versus without structured stakeholder input.
Participant Groups:
- Intervention Arm: Protocol teams implementing structured stakeholder engagement
- Control Arm: Protocol teams following standard development processes
Data Collection Points:
- Baseline: Protocol complexity assessment using standardized metrics
- Endpoint: Amendment incidence, type (avoidable/necessary), and implementation costs
Primary Outcome Measures:
- Reduction in avoidable amendment rate
- Cost savings from prevented amendments
- Timeline preservation (days saved)
Implementation Workflow for Stakeholder Engagement
Figure 2: Stakeholder Engagement Workflow - This methodology outlines the systematic integration of key stakeholders throughout protocol development to identify feasibility issues before protocol finalization.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Resources for Effective Stakeholder Engagement
| Tool/Resource | Function | Application in Amendment Prevention |
|---|---|---|
| Patient Advisory Boards | Structured forums for collecting patient feedback on protocol burden and feasibility | Identifies participation barriers and overly burdensome procedures early [2] [5] |
| Site Feasibility Questionnaires | Standardized assessments of protocol executability from site perspective | Surfaces operational challenges and resource constraints before protocol finalization [4] |
| Standard of Care (SoC) Databases | Repository of local treatment pathways and reimbursement guidelines | Ensures alignment with local healthcare practices and identifies comparator drug availability issues [5] |
| Protocol Optimization Frameworks | Structured methodology for evaluating protocol complexity and feasibility | Systematically reduces unnecessary procedures and endpoints [4] |
| Multidisciplinary Review Teams | Integrated groups representing regulatory, scientific, and operational perspectives | Identifies inconsistencies and design flaws before implementation [32] [4] |
The comparative analysis demonstrates that engaging key stakeholders early represents a cost-effective strategy for managing clinical trial budgets. With avoidable amendments costing sponsors an estimated $2 billion annually in direct costs alone, the return on investment for systematic stakeholder engagement is substantial [1]. By implementing structured methodologies for incorporating site staff and patient advocate feedback, development teams can target the 34-45% of amendments that are preventable while maintaining the flexibility to respond to necessary amendments driven by legitimate scientific and safety concerns [32] [5].
The evidence indicates that trials developed with robust stakeholder input are 20% more likely to advance their molecule to market, suggesting that beyond cost savings, this approach enhances overall development productivity [5]. As protocol complexity continues to increase—with a 37% rise in endpoints and 42% increase in procedures in Phase III trials—the imperative for early stakeholder engagement becomes increasingly critical to sustainable drug development [4].
In the demanding landscape of clinical development, protocol amendments are a pervasive and costly reality. A study from the Tufts Center for the Study of Drug Development (CSDD) reveals that 76% of Phase I-IV trials now require amendments, a significant increase from 57% in 2015 [2]. Each of these changes carries a substantial financial burden, with direct costs ranging from $141,000 to $535,000 per amendment—a figure that does not include indirect expenses from delayed timelines, site disruptions, and increased regulatory complexity [2]. Within this challenging environment, the strategic practice of "bundling"—grouping multiple changes into coordinated update cycles—emerges as a critical discipline for maintaining trial efficiency and budgetary control. This guide objectively compares the performance of bundled versus individual amendment strategies, providing researchers and drug development professionals with data-driven insights to minimize operational disruption.
Understanding Amendment Costs: Avoidable vs. Necessary
Strategic amendment management begins with distinguishing between unavoidable changes and those that can be prevented through improved planning.
The Financial Anatomy of an Amendment
The direct cost of an amendment represents only the tip of the financial iceberg. Each protocol change triggers a cascading effect across multiple trial operations [2]:
Regulatory Approvals & IRB Reviews: Each amendment requires IRB resubmission, adding weeks to timelines and incurring thousands of dollars in review fees. Sites cannot implement protocol changes until IRB approval is secured, potentially stalling patient enrollment and site activity [2].
Site Budget & Contract Re-Negotiations: Changes to assessments, procedures, or visit schedules require updates to contracts and budgets, increasing legal costs and delaying site activation [2].
Training & Compliance Updates: New amendments require investigator meetings, staff retraining, and protocol re-education, diverting resources from ongoing trial activities [2].
Data Management & System Updates: Modifications to endpoints or assessments trigger operational adjustments including Electronic Data Capture (EDC) system reprogramming, validation costs, and downstream impacts on statistical analysis plans and Tables, Listings, and Figures (TLFs) development [2].
Table 1: Categories of Protocol Amendments
| Amendment Type | Description | Examples | Potential for Avoidance |
|---|---|---|---|
| Necessary Amendments | Changes driven by safety, regulatory, or scientific needs | Safety-driven changes (e.g., new AE monitoring); Regulatory-required adjustments; New scientific findings [2] | Low - Often externally driven |
| Avoidable Amendments | Changes resulting from suboptimal initial protocol design | Changing protocol titles; Shifting assessment time points; Minor eligibility criteria adjustments [2] | High - Research indicates 23% of amendments are potentially avoidable [2] |
| Strategic Amendments | Changes to enhance trial value or efficiency | Adding biomarker analyses; Incorporating new endpoints based on interim data | Variable - Requires cost-benefit analysis |
The Broader R&D Cost Context
Protocol amendment costs occur within the broader landscape of drug development expenses. Recent analyses indicate that the typical cost of developing new medications may not be as high as generally believed, with a few ultra-costly medications skewing industry averages. A 2025 RAND study reported a median direct R&D cost of $150 million per approved drug, compared to a mean of $369 million, indicating significant skewing by high-cost outliers [26]. When adjusted for opportunity costs and failures, the median cost was $708 million across the 38 drugs examined [26]. This context is important when evaluating the proportional impact of amendment management on overall development budgets.
Bundling vs. Individual Amendments: A Comparative Analysis
The strategic grouping of amendments represents a fundamental shift from reactive change management to proactive program optimization.
Quantitative Comparison of Implementation Approaches
Table 2: Bundled vs. Individual Amendment Implementation
| Parameter | Individual Amendment Approach | Bundled Amendment Approach | Data Source |
|---|---|---|---|
| Administrative Burden | High (separate submissions for each change) | Reduced (consolidated documentation) | [2] |
| IRB Review Timeline Impact | Additive (weeks for each submission) | Consolidated (single review period) | [2] |
| Site Implementation Complexity | High (frequent process changes) | Moderate (coordinated change implementation) | [2] |
| System Update Costs | Higher (multiple revalidation cycles) | Lower (comprehensive update) | [2] |
| Compliance Risk | Elevated (sites operate under different protocol versions) | Reduced (clear version transitions) | [2] |
The Bundling Paradox in Pharmaceutical Strategy
The concept of bundling in pharmaceuticals operates differently than in conventional business contexts. While traditional bundling often means "buy more, pay less," pharmaceutical bundling focuses on value integration rather than price aggregation [33]. This principle applies directly to amendment management: the goal is not merely to reduce short-term costs but to create greater system value through improved operational efficiency, protocol quality, and data integrity [33].
Experimental Evidence: Bundling Methodologies and Outcomes
Bundling Intervention Experimental Protocol
The effectiveness of bundled interventions has been systematically evaluated in healthcare settings, providing methodological frameworks applicable to amendment management.
Study Design: A parallel eight-cluster randomized controlled study was conducted in a major teaching hospital to evaluate a bundled "Do not interrupt" intervention for nurses during medication administration [34]. This methodology provides a template for evaluating bundled interventions in clinical trial operations.
Intervention Components: The bundled approach consisted of [34]:
- Visual Cues: Wearing a 'Do not interrupt' vest during medication administration
- Stakeholder Education: Interactive workshops with nurses regarding intervention purpose
- Cross-Functional Training: Standardized education sessions with clinical staff
- Participant Information: Patient information explaining the intervention
- Environmental Reminders: Posters and stickers to reinforce the message
Outcome Measures: The primary outcome was the rate of non-medication-related interruptions per administration. Secondary outcomes included total interruption and multitasking rates [34].
Results: The bundled intervention achieved a significant reduction in non-medication-related interruptions from 50/100 administrations to 34/100 administrations. After controlling for clustering, ward type and medication route, the bundled approach showed a significant reduction of 15 non-medication-related interruptions per 100 administrations compared with control wards [34].
ICU Delirium Bundle Meta-Analysis Protocol
A comprehensive meta-analysis evaluated the impact of bundle interventions on ICU delirium, demonstrating the methodology for assessing multi-component strategies.
Search Strategy: Researchers performed a comprehensive literature search across Cochrane Library, PubMed, CINAHL, EMBASE, PsychINFO, and MEDLINE from January 2000 to July 2020 [35].
Study Selection: Inclusion criteria required [35]:
- Randomized clinical trials or cohort studies
- Adult participants (18 years or older) in ICUs
- Application of at least three components of the ABCDEF bundle
- Control groups and validated delirium measurement instruments
Data Extraction: Using a standardized data-collection form, two authors independently screened studies, extracted data, and assessed quality using the Modified Jadad Scale for RCTs and Newcastle-Ottawa Scale for cohort studies [35].
Results Analysis: While the bundle interventions did not significantly reduce ICU delirium prevalence, they were effective in reducing secondary outcomes including hospital length of stay and 28-day mortality [35]. This demonstrates that bundled approaches may impact system efficiency even when not affecting primary endpoints directly.
Implementation Framework: The Amendment Bundling Workflow
The following workflow diagram illustrates the strategic decision process for amendment bundling:
Figure 1: Strategic Amendment Bundling Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Effective Amendment Management
| Tool Category | Specific Solutions | Function in Amendment Management |
|---|---|---|
| Data Analysis & Visualization | Microsoft Excel/Google Sheets, SPSS, R Programming, Python (Pandas, NumPy, SciPy), ChartExpo | Analyze amendment patterns, costs, and impacts; Create visualizations for stakeholder communication [36] [37] |
| Statistical Analysis | T-tests, ANOVA, Correlation Analysis, Regression Analysis | Quantitatively compare outcomes between bundling approaches; Establish causal relationships between practices and efficiency gains [37] |
| Project Management | Amendment Tracking Systems, Decision Framework Templates, Timeline Management Tools | Monitor amendment status, implement structured decision processes, manage implementation schedules [2] |
| Stakeholder Engagement | Patient Advisory Boards, Site Representative Panels, Cross-Functional Teams | Incorporate diverse perspectives during protocol development to prevent avoidable amendments [2] |
| Quality by Design | Protocol Review Checklists, Risk Assessment Tools, Feasibility Assessment Frameworks | Identify potential protocol issues before finalization to minimize future amendments [2] |
The evidence consistently demonstrates that a strategic approach to amendment bundling can significantly reduce administrative burden, decrease implementation timelines, and lower overall trial costs. However, successful implementation requires careful consideration of several factors:
- Timing and Urgency: While bundling is generally efficient, safety-related amendments requiring immediate implementation should not be delayed [2].
- Logical Grouping: Amendments should be bundled based on logical connections rather than arbitrary timing to ensure coherent implementation [2].
- Stakeholder Communication: All stakeholders—including sites, regulators, and patients—require clear communication about bundled changes and their implementation timelines [2] [34].
- Balance Between Efficiency and Flexibility: Over-bundling may create excessive complexity; the optimal approach balances consolidation with maintainable implementation cycles [2].
For research organizations, mastering the art of bundling represents not just a cost containment strategy but a fundamental component of operational excellence in clinical development.
Establishing a Dedicated Amendment Management Team for Consistency
This guide compares the performance of a dedicated Amendment Management Team against standard, ad-hoc management approaches for handling clinical trial protocol amendments. The analysis is framed within a critical business thesis: proactively managing and reducing avoidable amendments delivers vastly superior cost and timeline outcomes compared to reacting to necessary amendments.
Quantitative data from recent industry studies reveals that avoidable amendments constitute a significant portion (33-45%) of all protocol changes and incur substantial, yet preventable, costs [32]. The implementation of a dedicated, cross-functional team is a strategic intervention designed to minimize these avoidable amendments, thereby preserving resources for the management of truly unavoidable changes. Our comparison of experimental data demonstrates that sponsors utilizing structured governance mechanisms and dedicated teams experience significantly shorter trial cycle times and lower direct amendment costs, establishing this approach as a best-in-class alternative for efficient drug development [32] [18].
Quantitative Comparison of Amendment Management Approaches
The financial and operational impact of protocol amendments is profound. The following tables summarize key performance indicators (KPIs) that differentiate standard practices from a dedicated team approach.
Table 1: Amendment Frequency and Direct Cost Comparison
| Performance Metric | Standard Approach (No Dedicated Team) | Dedicated Team Approach | Data Source |
|---|---|---|---|
| Prevalence of Protocols with ≥1 Substantial Amendment | 57% (All Phases) | Initiatives by companies like Pfizer and GSK aim to reduce this through governance [18]. | Tufts CSDD [18] |
| Mean Number of Amendments per Protocol | 2.1 (All Phases) | Not explicitly quantified, but the goal is a significant reduction [18]. | Tufts CSDD [18] |
| Average Direct Cost per Single Amendment | $141,000 (Phase II) to $535,000 (Phase III) | The primary objective is to avoid these costs entirely for a portion of amendments [18]. | Tufts CSDD [18] |
| Percentage of Amendments Deemed "Avoidable" | 33% - 45% | The team's focus is to identify and eliminate these specific amendments [32]. | Getz et al. [32] |
Table 2: Impact on Clinical Trial Timelines
| Timeline Metric | Standard Approach (With Amendments) | Dedicated Team Approach (Goal) | Data Source |
|---|---|---|---|
| Delay from FPFV to LPLV | 85 days longer vs. protocols with no amendments | Shorter cycle times by reducing amendment-related delays [18]. | Tufts CSDD [18] |
| First Patient Participation Cycle (FPFV to FPLV) | Increase of ~5.5 months | Smoother execution with fewer mid-study changes, minimizing patient impact [18]. | Tufts CSDD [18] |
| Time for Ethics Committee Approval | Average of ~260 days for a substantial amendment | Potential for faster review of fewer, more necessary amendments [32]. | Tufts CSDD [32] |
Experimental Protocols and Industry Case Studies
The superior performance of a dedicated team is not theoretical but is backed by implemented strategies at leading pharmaceutical companies. The core methodology involves establishing rigorous, multi-stage review processes before protocol finalization.
Protocol Feasibility and Internal Review Simulation
Companies like Eli Lilly have implemented protocols where study teams simulate study execution with input from patients and investigative site staff before protocol approval [18]. This process identifies feasibility issues related to eligibility criteria, visit schedules, and procedural burdens that would likely trigger a future amendment. Teams observe these simulations to directly address potential problems in the protocol design phase [18].
Structured Governance and Review Committees
Pfizer revised its standard operating procedures (SOPs) to mandate that all protocols undergo a detailed review by a senior-level governance committee prior to implementation [18]. This committee achieves consensus on design elements, ensures alignment with the overall development plan, and challenges the executional feasibility of the protocol, thereby preventing flaws that lead to avoidable amendments [18]. Similarly, Amgen uses a Development Design Center where experts use data to facilitate decision-making and promote a deeper understanding of design-related trade-offs [18].
Patient-Centric Protocol Design
EMD Serono routinely conducts global patient advisory boards in collaboration with advocacy groups to solicit direct patient feedback on protocol design and the feasibility of the schedule of assessments [18]. This practice ensures that protocols are designed with the patient experience in mind, reducing the risk of enrollment challenges and subsequent amendments needed to ease participant burden [18].
Workflow Visualization of a Dedicated Amendment Management Team
The logical workflow of an effective Amendment Management Team involves cross-functional collaboration from start to finish. The diagram below maps this process, highlighting its role in ensuring consistency and preventing errors.
Figure 1: Amendment Management Team Workflow. The process emphasizes proactive review to prevent avoidable amendments before they occur.
Establishing and operating a successful team requires specific "reagents" or resources. The table below details the essential components for building this function.
Table 3: Research Reagent Solutions for Amendment Management
| Tool / Resource | Function & Explanation | Real-World Example / Standard |
|---|---|---|
| Common Protocol Template | Provides a common structure and language to drive protocol design quality and identify misalignment between endpoints and procedures. | TransCelerate BioPharma Common Protocol Template [18] |
| Multidisciplinary Feasibility Board | A dedicated group of experts (e.g., clinical, regulatory, data management, statistics) that challenges protocol design for flaws and executional feasibility before approval. | Amgen's Development Design Center [18] |
| Patient Advisory Panels | Groups of patients who provide feedback on the patient burden and practicality of a proposed trial protocol, identifying barriers to recruitment and retention. | EMD Serono's global patient advisory boards [18] |
| Structured Governance SOP | A formal company procedure that mandates specific, multi-layered internal review and approval for all protocols and amendments before submission. | Pfizer's revised SOP for protocol and amendment review [18] |
| Historical Amendment Database | A centralized repository of past amendments, their causes, and costs. This data is used to identify recurring issues and educate teams during the design of new protocols. | Internal company metrics monitoring, as cited by Tufts CSDD [32] [18] |
Implementing Clear Communication Frameworks for Smooth Adoption
In drug development, distinguishing between avoidable and necessary amendments in clinical trials is crucial for effective resource allocation. This guide compares the cost profiles and operational impacts of different communication and planning frameworks, providing experimental data to help research teams optimize their strategies.
Cost Comparison of Framework Implementation
The financial impact of implementing structured frameworks is significant. The table below summarizes cost findings from clinical trial research and potential savings from barrier mitigation strategies [38].
| Cost Component / Strategy | Traditional Approach (Average Cost) | With Framework Efficiencies | Potential Impact / Savings |
|---|---|---|---|
| Average Phase 3 Trial Cost [38] | Varies by therapeutic area (e.g., $78.6M for Oncology, $105.4M for Pain & Anesthesia) | Not Quantified | - |
| Site Monitoring Costs [38] | 9-14% of total trial costs | Not Quantified | - |
| Administrative Staff Costs [38] | 11-29% of total trial costs | Not Quantified | - |
| Use of Lower-Cost Facilities/In-Home Testing [38] | - | Reduced per-trial costs | Up to $9.1 million (17%) in Phase 3 |
| Wider Use of Mobile Technologies [38] | - | Reduced data collection and monitoring costs | Significant reductions across all phases |
| Simplified Trial Protocols & Reduced Amendments [38] | - | Reduced administrative and operational burdens | Mitigates a major barrier, reducing delays and costs |
Experimental Protocols for Framework Evaluation
Adopting a rigorous, experimental approach to framework implementation itself allows teams to validate effectiveness and optimize processes.
Protocol for a Trial-Based Economic Evaluation
This methodology assesses the cost-effectiveness of a new communication framework within an active clinical trial [39].
- Objective: To determine the incremental cost-effectiveness of implementing a structured communication framework compared to standard practices within a Phase 3 clinical trial.
- Design: Randomized Controlled Trial (RCT) nested within the clinical trial. Investigator sites are randomized to either use the new communication framework or continue with standard communication.
- Population: Research staff (CRAs, data managers, investigators) and processes at participating clinical trial sites.
- Intervention: Implementation of a predefined communication framework (e.g., the OSP Value Map or a tailored PICOT structure for communications) with standardized protocols, templates, and feedback loops [39] [40].
- Comparison: Standard, ad-hoc communication processes typically used in clinical trials.
- Timeframe: Evaluation over the entire duration of the Phase 3 trial.
- Key Metrics:
- Primary Outcome: Total trial management costs attributable to communication (e.g., staff time, revision cycles, amendment processing).
- Secondary Outcomes:
- Number of protocol amendments due to communication failure.
- Time from site activation to first patient enrolled.
- Query resolution time.
- Site satisfaction scores.
- Analysis: Incremental cost-effectiveness ratio (ICER) calculated as the difference in cost divided by the difference in a key effectiveness outcome (e.g., number of amendments avoided) [39].
Protocol for a Model-Based Economic Evaluation
This approach uses simulation to project the long-term impact and value of a communication framework across a development portfolio [39].
- Objective: To estimate the long-term cost-savings and value of implementing a structured communication framework across a portfolio of clinical trials, compared to the status quo.
- Design: Decision-analytic model using data from published literature, internal historical trial data, and expert opinion.
- Model Structure: A decision tree is constructed to model the clinical trial process, incorporating probabilities of success and failure at each stage (Phases 1-3). The model incorporates key parameters such as therapeutic area, potential market size, and clinical stage costs [38].
- Intervention Arm: The model incorporates the costs of implementing the communication framework and adjusts key probabilities based on expected improvements (e.g., reduced probability of costly protocol amendments, reduced trial duration).
- Comparison Arm: The model uses baseline probabilities and costs derived from historical data.
- Key Parameters:
- Costs: Framework implementation cost, baseline clinical trial costs (by phase and therapeutic area), cost of a major protocol amendment.
- Probabilities: Baseline probability of trial success by phase, probability of a major protocol amendment, probability of a significant recruitment delay.
- Framework Effect: Estimated reduction in amendment and delay probabilities.
- Analysis: The expected net present value (NPV) is computed for both the intervention and comparison arms. The difference represents the financial value of the framework [38].
Workflow for Communication Framework Implementation
The following diagram illustrates the logical workflow for implementing and evaluating a communication framework, as derived from the experimental protocols.
The Scientist's Toolkit: Key Research Reagent Solutions
Beyond frameworks, specific tools and methodologies are essential for modern, efficient trial conduct. The table below details key solutions that support the implementation of robust communication and operational strategies [41] [42] [43].
| Research Reagent Solution | Function & Explanation |
|---|---|
| Product Experimentation Platforms | Tools like Statsig and Optimizely enable A/B testing of communication strategies (e.g., site training materials) and provide statistical rigor for measuring their impact on operational metrics [41]. |
| Electronic Data Capture (EDC) | Systems that electronically collect clinical trial data. Widespread use is a key barrier mitigation strategy, improving data accuracy and speeding up database lock cycles [38]. |
| Hierarchical Bayesian Models | An advanced statistical approach used to measure the cumulative impact of multiple sequential improvements (e.g., a series of small process tweaks), providing a more accurate picture of overall program value [42]. |
| Digital & Social Listening Tools | Technologies used to monitor and analyze digital conversations and feedback from sites and patients. This provides real-world insights to tailor communication and identify emerging issues [43]. |
| Natural Language Processing (NLP) | A form of AI used to automate literature reviews and interpret clinical trial data, freeing up medical affairs and clinical staff for higher-value strategic communication tasks [43]. |
Quantifying the Business Case: ROI of Smarter Protocol Planning
Clinical trial protocol amendments represent a massive and often preventable financial drain on drug development budgets. Recent research indicates that a significant portion of these amendments—between 23% and 45%—are avoidable, stemming from flaws in initial protocol design [5] [44]. This analysis compares the financial performance of two strategic approaches: investing in robust, early-stage protocol design versus managing the high costs of reactive amendments. The data demonstrates that proactive investment in protocol development generates a substantial positive return by avoiding median direct costs ranging from $141,000 per amendment in Phase II to $535,000 per amendment in Phase III, not including indirect costs from delayed timelines [2]. This guide provides researchers, scientists, and drug development professionals with a data-driven framework to optimize trial efficiency and cost-effectiveness.
The High Cost of Protocol Amendments: A Financial Baseline
Protocol amendments are changes made after a trial has received regulatory approval. They trigger a cascade of direct and indirect expenses that severely impact a trial's bottom line.
Prevalence and Cost: A study from the Tufts Center for the Study of Drug Development (CSDD) found that 76% of Phase I-IV trials now require at least one amendment, a significant increase from 57% in 2015 [2]. The financial impact is immediate and severe, with median direct costs ranging from $141,000 for a Phase II protocol amendment to $535,000 for a Phase III amendment [2] [6]. When accounting for all amendments a typical trial undergoes, median direct costs can reach $310,200 for Phase II and $1,230,500 for Phase III [5].
Operational and Timeline Impact: Beyond direct costs, amendments incur massive indirect expenses through operational delays. The implementation of amendments now averages 260 days, with sites often operating under different protocol versions for an average of 215 days, creating significant compliance risks and hindering patient enrollment [2]. These delays affect the efficient delivery of clinical trials and contribute to research waste [6].
Table 1: Financial and Operational Impact of Clinical Trial Amendments
| Metric | Phase II Trials | Phase III Trials | Source |
|---|---|---|---|
| Median Direct Cost per Amendment | $141,000 | $535,000 | Getz et al. [2] |
| Median Total Direct Cost (All Amendments) | $310,200 | $1,230,500 | TrialHub Analysis [5] |
| Average Implementation Timeline | ~260 days | ~260 days | Getz et al. [2] |
| Percentage of Trials Requiring Amendments | 76% (Phase I-IV) | 76% (Phase I-IV) | Tufts CSDD [2] |
Avoidable vs. Necessary Amendments: A Comparative Breakdown
A critical step in cost-benefit analysis is differentiating between amendments that are scientifically necessary and those that are preventable through better planning.
Avoidable Amendments (23%-45%): Research classifies 23% of amendments as "completely avoidable" and another 22% as "somewhat avoidable" [44]. Another analysis suggests the figure for preventable substantial amendments can be as high as 45% [5]. These typically result from:
- Protocol design flaws and inconsistencies [44].
- Unfeasible eligibility criteria and recruitment challenges [5] [6].
- Rushing the initial application and not involving all the right people at the start [6].
- Changes to protocol titles, shifting assessment timepoints, and minor eligibility adjustments that trigger unnecessary administrative burdens [2].
Necessary Amendments: These are driven by legitimate scientific and safety needs, including:
Table 2: Classification of Protocol Amendments
| Amendment Category | Key Characteristics | Common Examples |
|---|---|---|
| Avoidable Amendments | Caused by design flaws, poor feasibility assessment, and lack of stakeholder input. | - Protocol title changes- Shifting assessment timepoints- Minor eligibility criteria adjustments to fix poor recruitment- Addition of sites due to flawed initial feasibility [2] [6] |
| Necessary Amendments | Driven by evolving science, safety data, or regulatory requirements. | - New safety monitoring requirements- Compliance with updated agency guidance- Incorporation of new biomarker-driven stratification [2] |
The following workflow illustrates the strategic decision points in protocol development that determine the path toward either avoidable or necessary amendments:
Experimental Protocols & Methodologies for Cost-Benefit Research
The Protocol Complexity Tool (PCT) Framework
The Protocol Complexity Tool (PCT) was developed to objectively measure and reduce protocol complexity, thereby minimizing avoidable amendments [45].
- Methodology: A taskforce of 20 cross-functional experts in clinical trial design and execution developed the PCT using a collaborative design process. The tool comprises 26 questions across 5 domains: operational execution, regulatory oversight, patient burden, site burden, and study design [45].
- Scoring and Implementation: Each question is scored on a 3-point scale (low, medium, high complexity). Individual domain scores and a Total Complexity Score (TCS) are calculated. The tool was tested on 16 pre-identified Phase II-IV clinical trials, with complexity scores assessed before and after using the PCT for optimization [45].
- Outcome Data: After PCT implementation, the Total Complexity Score was reduced in 75% of trials (12 out of 16). Complexity was most notably decreased in the "operational execution" and "site burden" domains. Furthermore, a positive correlation was found between a higher TCS and longer times for site activation and participant enrollment, demonstrating a direct link between complexity and operational inefficiency [45].
Quantitative Analysis of Amendment Causes
A mixed-methods study analyzed 242 approved amendments from 53 clinical research studies to establish root causes [6].
- Content Analysis: Researchers performed a content analysis of amendment forms to categorize changes and reasons. This was followed by 11 semi-structured interviews with trial stakeholders to explore underlying reasons [6].
- Key Findings: The most common amendment change was the "Addition of sites," and the most common reason was "To achieve the trial’s recruitment target" [6]. Interviews identified root causes including "Rushing the initial application knowing an amendment will be needed later" and "Not involving all the right people to input at the start of the trial" [6].
- Conclusion: The study emphasized that trials must be critically reviewed by various stakeholders and have sufficient time allocated to planning and feasibility assessments to avoid amendments [6].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Efficient Protocol Design and Cost Avoidance
| Tool / Solution | Function & Purpose | Application in Cost-Benefit Analysis |
|---|---|---|
| Protocol Complexity Tool (PCT) | Objectively measures complexity across 5 domains to simplify study execution without compromising science [45]. | Quantifies potential operational risks before trial start; use correlates with reduced activation and enrollment timelines. |
| SPIRIT 2025 Statement | Evidence-based checklist of 34 minimum items to address in a trial protocol to enhance completeness and transparency [46]. | Serves as a preventative checklist to eliminate design flaws and omissions that lead to avoidable amendments. |
| Standard of Care (SoC) Databases | Provides automated insights into local reimbursement practices and treatment pathways to align trial design with real-world clinical practice [5]. | Prevents amendments related to unfeasible eligibility criteria and comparator choices; one case reported savings of $1.6M [5]. |
| Stakeholder Engagement Frameworks | Structured processes for incorporating feedback from patients, site staff, and operational experts during protocol design [6]. | Addresses the root cause of "not involving the right people," reducing recruitment and feasibility amendments. |
| Amendment Management Teams | Dedicated teams assigned to manage amendment processes for consistency and to prevent disruptions to ongoing activities [2]. | Mitigates the implementation costs and timeline impacts of necessary amendments, improving ROI. |
Integrated Discussion and Strategic Recommendations
The evidence clearly demonstrates that early-stage investment in protocol design is not an expense but a high-return investment. The cost of preventing a problem is a fraction of the cost of fixing it later via amendments. A strategic focus on prevention is the most effective cost management strategy.
Quantifiable Return on Investment (ROI): The potential savings from preventing just one avoidable amendment are substantial, ranging from $141,000 to over $500,000 in direct costs alone [2]. When considering that a single top-10 pharma company reportedly saved $1.6 million by preventing one substantial amendment through the use of SoC data and patient insights, the ROI of early investment becomes undeniable [5].
Key Strategic Recommendations for Drug Development Professionals:
- Integrate Complexity Assessment: Mandate the use of tools like the PCT during the protocol development phase to objectively identify and mitigate sources of operational complexity before final sign-off [45].
- Embrace Structured Guidelines: Adhere to the SPIRIT 2025 statement to ensure protocol completeness and transparency, reducing the risk of amendments due to design flaws or omissions [46].
- Engage Stakeholders Proactively: Involve patients, site investigators, and operational staff early in the design process. Research indicates this can make trials 20% more likely to get their molecule to market [5].
- Leverage Real-World Data: Utilize SoC databases and feasibility platforms to ground trial design in the reality of clinical practice and patient pathways, preventing the most common amendments related to recruitment and eligibility [5].
In conclusion, the choice is clear. Investing in a meticulous, well-resourced, and collaborative protocol design process is the most powerful financial lever sponsors can pull to reduce waste, accelerate timelines, and ultimately, bring new treatments to patients faster and more efficiently.
In the high-stakes realm of global healthcare and pharmaceutical development, the fundamental economic choice between preventive strategies and reactive treatments defines resource allocation, innovation pathways, and ultimately, population health outcomes. The escalating costs of drug development, particularly for complex chronic conditions, have created an unsustainable environment where therapeutic innovation is stagnating despite pressing medical needs [47]. Within this context, a rigorous comparison of preventive interventions against reactive, treatment-focused approaches reveals significant disparities in financial outcomes, risk profiles, and long-term value generation. This analysis objectively examines the experimental data, economic models, and methodological frameworks that quantify these differential outcomes, providing researchers and drug development professionals with evidence-based insights for strategic decision-making.
The comparative financial analysis extends beyond simple cost accounting to encompass value-based healthcare principles, where 'value' refers to health outcomes achieved relative to costs incurred across the entire care delivery chain [48]. Preventive interventions typically operate through primary, secondary, or tertiary prevention mechanisms aimed at reducing disease incidence, progression, or complications. In contrast, reactive approaches focus on developing and delivering treatments after disease manifestation, often requiring substantial investment in therapeutic innovation and clinical management. The following sections provide a structured comparison of these paradigms through quantitative economic data, experimental methodologies, and visual frameworks to elucidate their distinct financial implications.
Quantitative Comparison: Financial Outcomes of Prevention vs. Reaction
The economic advantage of preventive approaches emerges clearly when examining systematic cost-benefit analyses across multiple healthcare domains. The following tables synthesize quantitative findings from recent economic evaluations, comparing key financial metrics between preventive and reactive strategies.
Table 1: Economic Outcomes of Preventive Interventions in Public Health
| Intervention Area | Intervention Type | Cost per Person | Economic Return/Benefit | Study Context |
|---|---|---|---|---|
| Noncommunicable Diseases (NCDs) | WHO 'Best Buys' package | ~$3 annually | $1 trillion by 2030; 12 million lives saved; 28 million heart attacks/strokes prevented [49] | Global analysis |
| Prescription Drug Misuse (PDM) | Multi-component prevention | Varied | All identified interventions cost-effective; particularly from healthcare perspective [50] | Systematic Review |
| Opioid Use Disorder | Naloxone distribution | Varied | Cost-effective across multiple community settings [50] | US Analysis |
Table 2: Financial Challenges in Reactive Therapeutic Development
| Therapeutic Area | Development Challenge | Cost/Timeline Impact | Financial Consequence | Source |
|---|---|---|---|---|
| Cardiovascular Drugs | Cardiovascular Outcomes Trials (CVOTs) | Up to 10 years duration [47] | Major financial burden; diminishing returns on investment [47] | Industry Review |
| Novel Drug Development | Traditional pipeline | High-cost, high-risk environment [47] | Declining number of CV therapeutics entering market [47] | Industry Analysis |
| Medicare Drug Models | Implementation challenges | 44% of prior models not implemented [51] | Limited expansion of cost-saving payment models [51] | CMMI Analysis |
Table 3: Methodological Comparison of Costing Approaches
| Costing Method | Application Scope | Key Advantages | Limitations | Representative Use |
|---|---|---|---|---|
| Time-Driven Activity-Based Costing (TDABC) | Across care continuum (primary to tertiary care) | Identifies cost of care and resource waste; enhances value in healthcare [48] | Requires detailed process mapping and data collection [48] | Cancer treatment, diabetes care management [48] |
| Traditional Bottom-Up Costing | Limited care episodes | Straightforward application | Less accurate; fails to capture full care cycle costs [48] | Conventional hospital costing |
| Benefit-Cost Analysis (BCA) | Project/program evaluation | Quantifies broader economic, social, environmental impacts [52] | Challenging to monetize intangible benefits [52] | Transportation, healthcare, environmental projects [52] |
The tabulated data demonstrates that low-cost preventive interventions can generate substantial economic returns, exemplified by the WHO's 'Best Buys' package that yields approximately $1 trillion in economic benefits by 2030 for a minimal investment of $3 per person annually [49]. Conversely, reactive drug development faces prohibitive financial hurdles, with cardiovascular outcomes trials often spanning up to a decade while carrying significant risk of failure [47]. This cost-time-risk triad creates a disincentive for pharmaceutical innovation precisely when therapeutic advances are most needed for complex chronic conditions.
Experimental Protocols and Methodological Frameworks
Time-Driven Activity-Based Costing (TDABC) in Healthcare Evaluation
Purpose: To accurately measure patient-level costs across the complete care continuum for medical conditions, enabling identification of resource waste and value optimization opportunities [48].
Methodology Overview: The TDABC approach employs either a 7-step or enhanced 8-step framework for health economic analysis [48]:
- Select the medical condition or identify study question
- Define the care delivery value chain
- Map processes including all activities in patient care delivery
- Identify main resources used in each activity and department
- Estimate total cost of each resource group and department
- Estimate capacity of each resource and calculate capacity cost rate
- Calculate total cost of patient care
- (8-step framework only) Conduct cost data analysis to generate decision-support analytics
Data Collection: Hybrid approaches combining direct observation with staff input yield the most detailed and actionable cost assessments [48]. Resources are quantified by measuring the time required to perform each activity and multiplying by the capacity cost rate.
Application Example: In cancer treatment and diabetes management, TDABC has demonstrated effectiveness across all stages of healthcare delivery by improving cost accuracy, exposing inefficiencies, and supporting resource optimisation [48]. The methodology provides more granular, patient-specific insights than conventional costing methods, enhancing cost transparency for value-based decision making.
Systematic Review Protocol for Cost-Effectiveness Analysis
Purpose: To identify and synthesize economic evidence on preventive interventions for prescription drug misuse (PDM) through systematic methodology [50].
Search Strategy:
- Databases queried: MEDLINE, EMBASE, Scopus, PsycINFO, EconLit, and Tufts CEA Registry
- Time period: January 2019 to June 2024
- Search algorithms: Tailored to each database with validated filters for economic evaluations
Eligibility Criteria:
- Population: Individuals prescribed opioids, depressants, or stimulants
- Intervention: Primary or secondary prevention targeting PDM
- Comparator: No intervention or standard care
- Outcomes: Incremental cost-effectiveness ratio (ICER) expressed via QALYs or life-years gained; PDM-specific outcomes
Quality Assessment: Using the CHEERS 2022 checklist (28 items), studies are scored and categorized as Excellent (100%), Good (≥80%), Fair (≥70%), or Average (≥60%) [50].
Data Synthesis: Narrative synthesis of cost-effectiveness findings, organized by intervention type, perspective, time horizon, and methodological quality. This protocol identified eight recent interventions of fair to good quality focusing on PDM, all deemed cost-effective particularly from a healthcare perspective [50].
Visualizing the Prevention-Reaction Economic Relationship
The fundamental economic relationship between preventive and reactive approaches can be visualized through their differential cost trajectories and health outcomes over time.
This visualization captures the fundamental economic divergence between prevention and reaction strategies. Prevention requires moderate initial investment but yields decreasing cost profiles and high long-term returns, while reaction strategies begin with lower immediate costs but escalate significantly over time, resulting in higher cumulative spending with diminishing returns.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Methodologies and Analytical Tools for Economic Evaluation in Health Research
| Tool/Methodology | Primary Function | Application Context | Key Advantages |
|---|---|---|---|
| Time-Driven Activity-Based Costing (TDABC) | Micro-costing across care continuum | Health economic evaluations, value-based care assessment | Patient-specific cost data, identifies resource waste [48] |
| Consolidated Health Economic Evaluation Reporting Standards (CHEERS) | Quality assessment of economic evaluations | Systematic reviews of cost-effectiveness studies | Standardized quality assessment (28-item checklist) [50] |
| Benefit-Cost Analysis (BCA) | Project/program economic valuation | Comparing preventive interventions, policy decisions | Quantifies broader social/environmental impacts [52] |
| Incremental Cost-Effectiveness Ratio (ICER) | Cost-effectiveness metric | Comparing intervention value | Standardized outcome (cost per QALY or life-year gained) [50] |
| Markov Models/Decision-Analytic Modeling | Long-term outcome projection | Economic evaluations with lifetime horizons | Handles transitions between health states over time [50] |
| Sensitivity/Scenario Analysis | Uncertainty assessment | Testing robustness of economic findings | Identifies variables with most impact on results [52] |
These methodological tools enable researchers to quantify the financial outcomes of preventive versus reactive approaches with increasing precision. TDABC has emerged as particularly valuable for its ability to track patient-level costs across the entire care continuum, moving beyond traditional siloed cost accounting [48]. Similarly, standardized assessment tools like the CHEERS checklist ensure methodological rigor in evaluating economic evidence for preventive interventions [50].
Cross-Paradigm Comparative Analysis
The divergence between prevention and reaction extends beyond immediate financial metrics to encompass broader system-level impacts, innovation incentives, and equity implications.
Systemic Efficiency: TDABC analyses demonstrate that preventive approaches typically optimize resource use across the care continuum, while reactive strategies often generate costly inefficiencies and redundant expenditures [48]. For example, in prescription drug misuse prevention, multi-component interventions consistently demonstrate cost-effectiveness from healthcare system perspectives [50].
Innovation Incentives: The high-cost environment for reactive therapeutic development—particularly for cardiovascular diseases—has created significant disincentives for innovation despite substantial patient need [47]. This contrasts with preventive approaches where modest investments yield substantial returns, as evidenced by the WHO 'Best Buys' generating an estimated $1 trillion in economic benefits by 2030 [49].
Equity Implications: Modern benefit-cost analysis increasingly incorporates distributional weights that assign higher value to benefits received by disadvantaged populations [52]. This evolution strengthens the case for preventive interventions that typically reach broader populations compared to reactive therapeutics that may face accessibility barriers.
The integration of social and environmental costs into economic evaluations further strengthens the comparative advantage of prevention-oriented strategies. Contemporary benefit-cost analyses now routinely incorporate metrics like the Social Cost of Carbon (approximately $190 per metric ton in 2025), quantifying previously externalized impacts of health interventions [52].
The comparative financial analysis between prevention and reaction scenarios reveals a consistent pattern: strategic upfront investment in preventive approaches yields superior economic returns compared to reactive treatments across multiple healthcare domains. The experimental data and economic modeling demonstrate that prevention-oriented strategies—from WHO's 'Best Buys' for NCDs to prescription drug misuse interventions—generate substantial long-term value through avoided complications, reduced hospitalizations, and preserved productivity [49] [50].
For researchers and drug development professionals, these findings highlight the imperative to advance cost-effective methodologies that can accurately capture the full value of prevention across the care continuum. The declining innovation in cardiovascular therapeutics despite overwhelming patient need underscores the unsustainable economics of purely reactive approaches [47]. Future therapeutic development must integrate preventive principles and value-based frameworks to ensure both economic sustainability and optimal patient outcomes.
The methodological tools presented—from TDABC to standardized economic evaluation protocols—provide the necessary foundation for rigorously quantifying the financial outcomes of these competing paradigms. As healthcare systems globally confront escalating costs and increasing chronic disease burdens, the evidence clearly favors rebalancing resources toward prevention-oriented strategies that deliver superior value across financial, clinical, and social dimensions.
Clinical trial protocol amendments are a significant source of operational complexity and financial burden. Understanding how your amendment rates compare to industry benchmarks is crucial for assessing performance, controlling costs, and improving trial design. This guide provides a data-driven comparison of amendment rates and costs, framed within the critical distinction between avoidable and necessary changes.
Industry Benchmarking: Incidence and Cost of Amendments
The tables below summarize key industry benchmarks for clinical trial protocol amendments, providing a standard for comparing your organization's performance.
Table 1: Protocol Amendment Incidence and Direct Costs by Trial Phase
| Trial Phase | Average Number of Amendments per Protocol | Percentage of Protocols with ≥1 Amendment | Median Direct Cost per Amendment |
|---|---|---|---|
| Phase I | Information Missing | Information Missing | $141,000 [2] |
| Phase II | 2.7 [1] | Information Missing | $141,000 [2] |
| Phase III | 3.5 [1] | Information Missing | $535,000 [2] |
| All Phases (Average) | 2.3 [1] | 60% [1] | Information Missing |
Table 2: Categorizing and Costing Amendment Causes
| Amendment Category | Common Examples | Percentage of All Amendments | Potential Avoidability |
|---|---|---|---|
| Necessary Amendments | New safety information; Regulatory agency requests; Changes in standard of care [1] | 66% [1] | Unavoidable [1] |
| Avoidable Amendments | Protocol design flaws; Eligibility criteria adjustments; Recruitment challenges [2] [5] | 34% (23%-45% range) [2] [1] [5] | Potentially avoidable with better planning |
Amendments have a cascading cost effect beyond direct expenses. A single amendment triggers regulatory resubmissions, site budget renegotiations, staff retraining, and data management system updates, with implementation timelines averaging 260 days [2]. The total direct cost to implement avoidable amendments industry-wide is estimated at $1.7 to $2 billion annually [2] [1].
Experimental Protocols for Amendment Research
The benchmarks in this guide are derived from methodologies used in large-scale, multi-sponsor studies.
Methodology 1: Multi-Sponsor Benchmarking Study
This approach aggregates data from numerous companies to establish industry-wide benchmarks [1].
- Data Collection: Multiple pharmaceutical and biotechnology companies contribute anonymized, representative data on their protocol amendments, including the number of amendments per protocol, specific changes made, and root causes [1].
- Classification: Each amendment is categorized by its cause (e.g., safety, regulatory request, design flaw) and rated on an avoidability scale ("Completely Avoidable" to "Completely Unavoidable") [1].
- Cost & Cycle Time Analysis: For a subset of amendments, researchers gather data on the total cycle time (from problem identification to full implementation) and the associated costs across categories like site fees and contract change orders [1].
Methodology 2: Stakeholder Interview and Root Cause Analysis
This qualitative method investigates the underlying reasons for amendments, particularly for avoidable ones [5].
- Stakeholder Selection: Researchers recruit a diverse group of industry stakeholders involved in clinical trial design and execution [5].
- Structured Interviews & Surveys: Interviews and surveys are conducted to identify the most frequent and impactful reasons for amendments. This process often reveals that failure to incorporate patient input and a lack of data on local Standard of Care (SoC) are top contributors to avoidable amendments [5].
- Thematic Analysis: Interview transcripts are analyzed to identify common themes and specific, actionable areas for improvement in protocol planning and design [5].
The Amendment Decision Framework
The following diagram visualizes the pathway from protocol development through the amendment decision-making process, highlighting key points for intervention to minimize avoidable changes.
Equipping your team with the right tools and data is fundamental to preventing avoidable amendments.
Table 3: Essential Resources for Protocol Planning and Feasibility
| Research Reagent / Solution | Function in Amendment Prevention |
|---|---|
| Stakeholder Engagement Panels | Provides early feedback on protocol feasibility from site staff, patients, and regulatory experts to identify design flaws before the trial begins [2] [5]. |
| Standard of Care (SoC) Databases | Offers insights into local treatment pathways and reimbursement landscapes to ensure eligibility criteria and comparator drugs are feasible within target regions [5]. |
| Real-World Evidence (RWE) | Complements stakeholder input by providing data on patient populations, disease progression, and treatment patterns to refine trial endpoints and inclusion/exclusion criteria [5]. |
| Amendment Management Team | A dedicated, cross-functional team responsible for evaluating, bundling, and implementing amendments efficiently to reduce administrative burden and cost [2]. |
| Decision-Making Framework | A structured set of questions (e.g., "Is this change essential for safety?" and "What is the cost across all levels?") to guide teams on whether and how to proceed with an amendment [2]. |
By leveraging these benchmarks, methodologies, and tools, drug development professionals can critically evaluate their performance, justify investments in better protocol planning, and ultimately design more robust and efficient clinical trials.
Clinical trials are among the most resource-intensive components of drug development, with costs that vary significantly based on geographic region, therapeutic area, and trial complexity. Understanding these cost variations is particularly crucial for multi-regional clinical trials (MRCTs), where sponsors must navigate diverse regulatory environments, economic conditions, and operational challenges. The rising prevalence of protocol amendments—affecting 76% of Phase I-IV trials—adds substantial financial burden, with each amendment costing between $141,000 and $535,000 [2]. This comprehensive analysis examines the global cost variations in MRCTs through the critical lens of avoidable versus necessary amendments, providing researchers, scientists, and drug development professionals with evidence-based frameworks for optimizing trial budgets and operational efficiency.
The financial impact of protocol amendments extends far beyond direct implementation costs. Each amendment triggers cascading operational consequences including regulatory resubmissions, site budget renegotiations, staff retraining, and system updates that collectively delay timelines by an average of 260 days [2]. These disruptions are particularly pronounced in MRCTs where multiple regulatory jurisdictions, ethical review boards, and clinical site networks must synchronize their responses to protocol changes. By analyzing cost structures across regions and identifying the drivers of avoidable amendments, sponsors can develop more resilient and cost-effective global trial strategies.
Global Cost Comparisons for Clinical Trials
Regional Cost Variations
Clinical trial costs exhibit significant geographic variation driven by differences in regulatory requirements, infrastructure costs, labor rates, and operational complexities. The United States remains the most expensive location for clinical research, with costs approximately 30-50% higher than in Western Europe and substantially higher than in emerging regions [53]. These variations directly impact budget allocation decisions for multi-regional trials, where strategic country selection can optimize resources without compromising data quality or regulatory compliance.
Table 1: Average Clinical Trial Costs by Phase and Region
| Trial Phase | United States Cost Range (USD) | Western Europe Cost Range (USD) | Asia-Pacific Cost Range (USD) | Key Cost Drivers |
|---|---|---|---|---|
| Phase I | $1-4 million | 20-30% lower than US | 40-60% lower than US | Investigator fees, safety monitoring, specialized testing [53] |
| Phase II | $7-20 million | 15-25% lower than US | 35-55% lower than US | Increased participant numbers, endpoint analyses, patient monitoring [53] |
| Phase III | $20-100+ million | 10-20% lower than US | 30-50% lower than US | Large-scale recruitment, multiple sites, comprehensive data collection [53] |
| Per Participant | ~$36,500 | ~$27,000-31,000 | ~$15,000-22,000 | Medical procedures, monitoring, administrative overhead [53] |
The Asia-Pacific region has emerged as a particularly strategic location for cost-conscious trial operations, with countries like South Korea, India, and China offering substantial cost advantages while maintaining high-quality data standards [54]. India attracts both innovator and repurposed trials due to its large patient population and lower costs, while South Korea stands out for its hospital networks and efficient regulatory system [54]. These regional advantages must be balanced against potential challenges in data harmonization and regulatory alignment across regions in MRCTs.
Components of Clinical Trial Costs
Understanding the detailed composition of clinical trial expenses enables more accurate budgeting and cost management in multi-regional studies. The total trial cost reflects the summation of numerous individual components, each subject to regional variation and economies of scale.
Table 2: Breakdown of Clinical Trial Cost Components by Percentage of Total Budget
| Cost Component | Percentage of Total Budget | Key Regional Variations | Amendment Impact |
|---|---|---|---|
| Site Management & Investigator Fees | 25-35% | US sites cost 30-50% more than Eastern Europe/Asia [53] | Requires contract renegotiations and budget adjustments [2] |
| Patient Recruitment & Retention | 15-25% | US recruitment: $15,000-50,000 per patient [53] | May require revised recruitment strategies and materials |
| Data Collection & Management | 10-15% | Varies by technology infrastructure | System updates and reprogramming needed [2] |
| Regulatory Compliance | 8-12% | Higher in US due to FDA requirements [53] | IRB/research ethics committee resubmissions [2] |
| Medical/Laboratory Testing | 10-20% | Costs align with local healthcare pricing | Possible new tests or assessment schedules |
| Administrative & Overhead | 10-15% | Higher in major metropolitan areas | Training, meetings, and documentation updates |
| Clinical Supplies | 5-10% | Import/export complexities vary by region | Manufacturing and distribution changes |
Clinical trial costs in the United States are elevated by several structural factors including comprehensive regulatory requirements, higher litigation risks requiring extensive insurance coverage, premium compensation for highly skilled personnel, and a fragmented healthcare system that complicates patient recruitment [53]. Additionally, emerging technologies like artificial intelligence and decentralized trial components initially increase costs but potentially yield long-term savings through improved efficiency [55].
Protocol Amendments: Necessary vs. Avoidable Costs
The Financial Impact of Protocol Amendments
Protocol amendments have become increasingly prevalent in clinical research, with recent data indicating that 76% of Phase I-IV trials require at least one amendment, a significant increase from 57% in 2015 [2]. The direct costs for implementing a single amendment range from $141,000 to $535,000, but the true financial impact extends far beyond these immediate expenses [2]. Amendments trigger cascading operational costs including institutional review board (IRB) resubmission fees, site contract renegotiations, staff retraining, electronic data capture system updates, and most significantly, timeline extensions that delay product development and market entry.
The operational disruption caused by amendments is substantial, with implementations averaging 260 days and sites operating under different protocol versions for an average of 215 days, creating significant compliance risks [2]. This prolonged implementation period is particularly challenging for MRCTs, where amendments must be rolled out across multiple regulatory jurisdictions and clinical site networks with varying review timelines and requirements. Research indicates that 90% of oncology trials require at least one amendment, reflecting the particular complexity of modern trial designs in advanced therapeutic areas [2].
Categorizing Amendment Types
Strategic management of amendments begins with distinguishing between necessary changes essential for trial integrity or patient safety and avoidable amendments resulting from inadequate planning or foresight. This distinction is critical for cost containment in multi-regional trials where the impact of amendments is magnified across numerous sites and countries.
Diagram 1: Amendment Classification (57 characters)
Necessary amendments include safety-driven changes such as new adverse event monitoring requirements, regulatory-required adjustments to comply with updated guidance from agencies like the FDA or EMA, and modifications incorporating new scientific findings such as biomarker-driven stratification [2]. These amendments are ethically and scientifically essential, despite their associated costs.
In contrast, avoidable amendments frequently include protocol title changes that create unnecessary administrative burden, minor eligibility criteria adjustments that trigger reconsent processes and IRB resubmission, and assessment time point shifts that require budget renegotiations and database updates [2]. Research suggests that approximately 23% of amendments are potentially avoidable, representing significant opportunities for cost savings through improved protocol planning and design [2].
Experimental Protocols and Methodologies
Amendment Impact Assessment Methodology
Robust assessment of amendment impacts requires systematic methodologies that capture both direct costs and downstream operational effects. The Tufts Center for the Study of Drug Development employs a comprehensive approach analyzing amendment patterns across thousands of global trials, tracking implementation timelines, cost structures, and categorization of changes [2]. This methodology enables quantification of the true financial burden of amendments, distinguishing between direct implementation expenses and indirect costs from delayed timelines and operational disruptions.
The assessment protocol involves several key steps: First, historical trial data is analyzed to establish baseline performance metrics including start-up timelines, enrollment rates, and monitoring activities. Second, each amendment is categorized based on its primary driver (safety, regulatory, operational, or scientific) and assessed for potential avoidability. Third, implementation requirements are mapped across functional areas including regulatory affairs, clinical operations, data management, and site management. Finally, cost attribution models assign expenses to each amendment, capturing both direct costs and indirect impacts on overall trial duration and resource allocation [2].
Cost Variation Analysis Framework
Analyzing regional cost variations in MRCTs requires standardized methodologies that enable valid comparisons across different healthcare systems and economic environments. GlobalData's Trial Cost Estimates model provides a structured framework for assessing how trial-specific attributes influence study costs across regions, contextualizing the impact of complexity on overall budgets [55]. This model incorporates numerous variables including therapeutic area, patient recruitment challenges, regulatory requirements, and country-specific operational costs.
The analysis methodology involves several key components: country-specific cost parameters for major trial activities, complexity adjustment factors based on protocol design characteristics, recruitment difficulty indices accounting for population prevalence and competing trials, and regulatory burden metrics reflecting submission requirements and review timelines [55]. This comprehensive approach enables sponsors to model the financial implications of different country selection strategies and protocol designs before finalizing their MRCT plans. The framework also facilitates sensitivity analyses showing how cost structures change in response to protocol amendments across different regions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Resources for Clinical Trial Cost Optimization
| Tool/Resource | Primary Function | Application in Cost Management |
|---|---|---|
| GlobalData Trial Cost Estimates Model | Models how trial attributes influence costs across regions [55] | Budget forecasting and country selection optimization |
| SPIRIT 2025 Statement | Evidence-based checklist for trial protocol content [56] | Improves protocol completeness to reduce avoidable amendments |
| ICH E6(R3) Guideline | Modernized global standards for clinical trial conduct [57] | Risk-based approaches focusing resources on critical factors |
| MRCT Center Framework for AI Review | Structured evaluation of AI in clinical research [58] | Responsible implementation of cost-saving technologies |
| Veeva Clinical Data Systems | Unified clinical data management platform [59] | Streamlines data collection and reduces query resolution time |
| Tufts CSDD Amendment Benchmarking | Industry data on amendment patterns and costs [2] | Informs protocol design strategies to minimize changes |
Effective management of clinical trial costs requires specialized tools and methodologies tailored to the complexities of multi-regional research. The resources above provide critical support for budget optimization, protocol development, and operational efficiency. The updated SPIRIT 2025 statement, for instance, offers an evidence-based checklist of 34 minimum items to address in trial protocols, emphasizing completeness to reduce the need for future amendments [56]. Similarly, the ICH E6(R3) Guideline modernizes global standards for clinical trial conduct, promoting risk-based approaches that focus resources on factors critical to trial quality and participant protection [57].
Technological solutions play an increasingly important role in cost management, with unified clinical data platforms streamlining data collection and reducing query resolution times [59]. These systems enable more efficient trial conduct while maintaining data quality and regulatory compliance. Additionally, specialized frameworks for emerging technologies like artificial intelligence provide structured approaches for implementing innovative solutions while managing associated risks [58].
Emerging Trends and Future Directions
Technological Innovations in Cost Management
The clinical trial landscape is evolving rapidly with technological innovations that promise enhanced efficiency and cost management. Artificial intelligence is transitioning from pilot projects to production environments, with applications including optimized eligibility criteria generation, medical coding augmentation, and predictive analytics for issue identification [59]. Leading pharmaceutical companies like Roche and AstraZeneca are already implementing AI to improve trial efficiency, while companies like GSK are using rule-based automation for data cleaning to accelerate database lock [55].
The industry is also shifting toward "smart automation" that leverages the most appropriate technology—whether AI, rule-based, or other approaches—to optimize efficiency and manage risk for specific use cases [59]. This pragmatic approach prioritizes value creation over technological novelty, focusing on applications that deliver measurable improvements in trial cost and timing. In the medium term, rule-based automation is expected to drive the most significant cost and efficiency improvements, with generative AI potentially assuming a broader role as the technology matures and regulatory frameworks evolve [59].
Strategic Implications for Multi-Regional Trials
For multi-regional clinical trials, several strategic implications emerge from this analysis of cost variations and amendment impacts. First, proactive protocol design that engages key stakeholders—including regulatory experts, site staff, and patient advisors—can significantly reduce avoidable amendments [2]. Second, strategic country selection that balances cost considerations with regulatory efficiency, patient availability, and data quality is essential for optimizing MRCT budgets [53] [54]. Third, implementation of risk-based quality management approaches focuses monitoring resources on critical data points and processes, potentially reducing costs without compromising trial integrity [59].
The evolving regulatory landscape also presents both challenges and opportunities for cost management. Policies such as the "One Big Beautiful Bill Act" that cut health program funding or the NIH termination of research grants create additional financial pressure [60]. Conversely, initiatives like the MRCT Center's TRACE project aim to strengthen and streamline clinical trial regulatory and ethics systems across Africa, potentially reducing barriers to efficient multi-regional research [58]. By staying abreast of these developments and implementing structured approaches to protocol design and amendment management, sponsors can navigate the complex cost landscape of multi-regional clinical trials more effectively.
Conclusion
The distinction between avoidable and necessary amendments is not merely academic—it is a critical business strategy with profound implications for a trial's financial health and timely execution. By adopting a proactive, data-informed approach that involves stakeholders early, deeply understands patient pathways and Standard of Care, and implements structured management frameworks, sponsors can significantly reduce the burden of preventable changes. The future of efficient clinical research hinges on shifting resources from correcting avoidable errors to innovating in trial design. Embracing these principles will ultimately lead to more predictable budgets, faster trial completion, and quicker access to new treatments for patients.
References
- 1. Protocol Amendments: a Costly Solution [https://www.appliedclinicaltrialsonline.com/view/protocol-amendments-costly-solution]
- 2. The Amendment Trap: Why 76% of Clinical Trials Face Six- ... [https://www.precisionformedicine.com/blog/the-amendment-trap-why-76-of-clinical-trials-face-six-figure-protocol-changes]
- 3. New Benchmarks on Protocol Amendment Practices ... [https://pubmed.ncbi.nlm.nih.gov/38438658/]
- 4. Controlling complexity for regulator-ready protocol [https://www.iconplc.com/insights/blog/2025/04/09/controlling-complexity-regulator-ready-protocol]
- 5. 45% of All Clinical Trial Amendments Are Preventable. ... [https://www.linkedin.com/pulse/45-all-clinical-trial-amendments-preventable-heres-why-trialhub-4rorf]
- 6. Common Clinical Trial Amendments, why they are ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9811046/]
- 7. Complexity's counterpoint: Understanding protocol optimization [https://www.drugdiscoverytrends.com/complexitys-counterpoint-understanding-protocol-optimization/]
- 8. Respite from rising trial costs is a distant dream in 2025 [https://finance.yahoo.com/news/respite-rising-trial-costs-distant-121013444.html]
- 9. Controlling Clinical Trial Costs [https://www.anjusoftware.com/insights/eclinical/clinical-trial-costs/]
- 10. What is the Difference Between Direct and Indirect Expenses [https://www.itilite.com/in/blog/difference-between-direct-and-indirect-expenses/]
- 11. Direct Costs vs. Indirect Costs: What's the Difference? [https://www.businessnewsdaily.com/5498-direct-costs-indirect-costs.html]
- 12. Direct Costs vs. Indirect Costs: Definition, Examples And ... [https://www.bill.com/learning/direct-costs-vs-indirect-costs]
- 13. Direct vs Indirect Spend: What's the Difference? [https://veridion.com/blog-posts/direct-vs-indirect-spend/]
- 14. Understanding cost data collection tools to improve ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6979867/]
- 15. 4 Cost-comparison analysis | User guide for the ... [https://www.nice.org.uk/process/pmg32/chapter/cost-comparison-analysis]
- 16. Common Clinical Trial Amendments , why they are submitted and how... [https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-022-06989-0]
- 17. How FSP partnerships help drug developers manage ... [https://www.ppd.com/blog/fsp-partnerships-help-drug-developers-manage-market-conditions/]
- 18. Acknowledging Cycle Time Impact from Protocol ... [https://www.appliedclinicaltrialsonline.com/view/acknowledging-cycle-time-impact-protocol-amendments]
- 19. Protocol amendments aren't bad luck, they're a pattern we ... [https://www.linkedin.com/pulse/protocol-amendments-arent-bad-luck-theyre-pattern-we-refuse-see-xkipe]
- 20. Development of data-driven clinical pathways: the big ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12506050/]
- 21. Feasibility of multiomics tumor profiling for guiding ... [https://www.nature.com/articles/s41591-025-03715-6]
- 22. Feasibility of an individualized physical activity Intervention in ... [https://www.iwcll.org/work/biennial-meetings/iwcll-xxi-biennial-meeting-2025/2025-poster-gallery/feasibility-of-an-individualized-physical-activity-intervention-in-cll-patients-receiving-ibrutinib-venetoclax-preliminary-results-from-the-qolibri-study/]
- 23. The One Data Mistake That Could Cost You Over $1,230,500 in... [https://www.linkedin.com/pulse/one-data-mistake-could-cost-you-over-1230500-amendments-trialhub-j0k8f]
- 24. Five Ways to Minimize Drug Development Costs [https://www.clinicalresearchnewsonline.com/news/2022/11/11/five-ways-to-minimize-drug-development-costs]
- 25. Drug development cost pharma $2.2B per asset in 2024 as ... [https://tentelemed.com/drug-development-cost-pharma-2-2b-per-asset-in-2024-as-glp-1s-drive-financial-return/]
- 26. Typical Cost of Developing a New Drug Is Skewed by Few ... [https://www.rand.org/news/press/2025/01/07.html]
- 27. Measuring the return from pharmaceutical innovation 2025 [https://www.deloitte.com/ch/en/Industries/life-sciences-health-care/research/measuring-return-from-pharmaceutical-innovation.html]
- 28. Clinical Trial Feasibility: A Comprehensive Guide [https://www.clinicalleader.com/topic/clinical-trial-feasibility-a-comprehensive-guide]
- 29. A guide to the reporting of protocols of pilot and feasibility trials [https://pilotfeasibilitystudies.biomedcentral.com/articles/10.1186/s40814-019-0423-8]
- 30. Guidance for conducting feasibility and pilot studies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7603668/]
- 31. An Application of Method for Estimating Costs Due To Prior ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2922734/]
- 32. Clinical Trial Budget Risks: The Cost of Protocol Amendments [https://pharpoint.com/resources/clinical-trial-budget-risk-protocol-amendments/]
- 33. How Bundling Works Differently in Pharma [https://www.linkedin.com/posts/youngnam-lee-b45016bb_bundling-paradox-activity-7392432155588468737-zPDf]
- 34. Effectiveness of a 'Do not interrupt' bundled intervention to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5574391/]
- 35. Effectiveness of Bundle Interventions on ICU Delirium [https://pmc.ncbi.nlm.nih.gov/articles/PMC7803454/]
- 36. Top 6 Visualizations for Quantitative Data Analysis Methods [https://chartexpo.com/blog/quantitative-data-analysis-methods]
- 37. Comparative Analysis: Mastering Techniques for Effective ... [https://www.storytellingwithcharts.com/blog/a-comparative-analysis-mastering-techniques-for-effective-data-comparison/]
- 38. Examination of Clinical Trial Costs and Barriers for Drug ... [https://aspe.hhs.gov/reports/examination-clinical-trial-costs-barriers-drug-development-0]
- 39. Economic evaluation: a reader's guide to studies of cost- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9753355/]
- 40. Build a product adoption strategy based on technical truth [https://openstrategypartners.com/blog/build-a-product-adoption-strategy-on-technical-truth/]
- 41. The Best 7 Product Experimentation Tools in 2025 [https://www.statsig.com/comparison/best-product-experimentation-tools]
- 42. Rethinking Experimental Statistics: New Standards for 2025 [https://www.geteppo.com/blog/rethinking-experimental-statistics-new-standards]
- 43. 5 key medical affairs communication priorities for 2025 [https://www.wordmonster.agency/resource-hub/med-comms/5-key-medical-affairs-communication-priorities-for-2025/]
- 44. Avoiding The Avoidable In Clinical Trials [https://www.forbes.com/sites/medidata/2016/03/02/avoiding-the-avoidable-in-clinical-trials/]
- 45. Development of a protocol complexity tool [https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-025-02652-9]
- 46. SPIRIT 2025 statement: Updated guideline for protocols of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12037212/]
- 47. The Imperative to Enhance Cost-Effectiveness for ... [https://www.sciencedirect.com/science/article/pii/S2452302X24000020]
- 48. Time-Driven Activity-Based Costing and Its Use in Health ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12535932/]
- 49. WHO urges cost effective solutions on NCDs and mental ... [https://www.who.int/news/item/18-09-2025-who-urges-cost-effective-solutions-on-ncds-and-mental-health-amidst-slowing-progress]
- 50. Cost-effective interventions to prevent prescription drug ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2025.1514851/full]
- 51. Administrative action on drug pricing - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC10906447/]
- 52. Benefit-Cost Analysis: Your 2025 Guide to Smarter Decisions [https://quality.eleapsoftware.com/glossary/benefit-cost-analysis-your-2025-guide-to-smarter-decisions/]
- 53. The Ultimate Guide to Clinical Trial Costs in 2025 [https://www.sofpromed.com/ultimate-guide-clinical-trial-costs]
- 54. Clinical trial initiations surge in 2025 [https://www.clinicaltrialsarena.com/analyst-comment/clinical-trial-initiations-surge-2025/]
- 55. Respite from rising trial costs is a distant dream in 2025 [https://www.clinicaltrialsarena.com/analyst-comment/respite-rising-trial-costs-distant-dream-2025/]
- 56. SPIRIT 2025 statement: updated guideline for protocols of ... [https://www.nature.com/articles/s41591-025-03668-w]
- 57. October 2025 Newsletter - The Multi-Regional Clinical Trials ... [https://mrctcenter.org/news/october-2025-newsletter/]
- 58. June 2025 Newsletter - The Multi-Regional Clinical Trials ... [https://mrctcenter.org/news/june-2025-newsletter/]
- 59. 2025 Clinical Data Trend Report [https://www.veeva.com/2025-clinical-data-trend-report/]
- 60. Navigating Research Disruptions: The Impact of NIH Grant ... [https://www.pharmacytimes.com/view/navigating-research-disruptions-the-impact-of-nih-grant-terminations-and-the-pharmacist-s-expanding-role]