Navigating Ethical Challenges in Pediatric Research Consent: A Comprehensive Guide for Researchers
This article provides a comprehensive analysis of the ethical challenges inherent in obtaining informed consent in pediatric research.
Navigating Ethical Challenges in Pediatric Research Consent: A Comprehensive Guide for Researchers
Abstract
This article provides a comprehensive analysis of the ethical challenges inherent in obtaining informed consent in pediatric research. Tailored for researchers, scientists, and drug development professionals, it explores the foundational ethical and regulatory frameworks, details practical methodologies for consent and assent processes, offers solutions for common implementation hurdles, and validates approaches through real-world case studies and international perspectives. The synthesis aims to equip professionals with the knowledge to conduct ethically sound and compliant pediatric research that respects the rights and welfare of child participants.
The Bedrock of Ethics: Principles and Regulations Governing Pediatric Consent
The evolution of international research ethics, from the Nuremberg Code to the Belmont Report, represents a foundational framework that all researchers must understand to conduct ethically sound studies, particularly when involving vulnerable populations like children. These documents emerged in response to historical ethical failures and established the core principles that underpin modern research regulations. For pediatric research, this framework necessitates special considerations, as children cannot provide independent informed consent, requiring researchers to navigate complex issues of parental permission, child assent, and risk assessment tailored to vulnerable populations. This technical support guide examines these historical documents and provides practical troubleshooting guidance for addressing common ethical challenges in pediatric research.
Foundational Documents & Their Core Principles
The Nuremberg Code (1947)
Historical Context: Developed in 1947 during the Nuremberg Military Tribunal where 23 Nazi doctors were accused of conducting brutal experiments on humans in concentration camps during World War II [1] [2]. The Code was created by American judges as a standard to judge these unethical practices and represents the first major international document to provide explicit guidelines on research ethics [3].
Key Principles:
- Voluntary consent is absolutely essential [1] [4]
- Research must yield fruitful results for the good of society unprocurable by other methods [4]
- Research should be based on animal experimentation and knowledge of the disease [2]
- Research must avoid all unnecessary physical and mental suffering [1]
- No experiment should be conducted where death or disabling injury is anticipated [4]
- The degree of risk should never exceed the humanitarian importance of the problem [2]
- Proper preparations and facilities must be provided to protect subjects [4]
- Experiments must be conducted by scientifically qualified persons [1]
- Human subjects should be at liberty to end the experiment at any time [4]
- The scientist must be prepared to terminate the experiment if continuation is likely to result in injury, disability, or death [2]
The Declaration of Helsinki (1964)
Historical Context: Adopted by the World Medical Association in 1964 as ethical principles for medical research involving human subjects, with several revisions since, most notably in 1975, 1983, 1989, 1996, and 2000 [1] [3]. It was developed by physicians appalled by the atrocities revealed at the Nuremberg Trials [1].
Key Advancements:
- Emphasized the distinction between research combined with professional care and non-therapeutic research [1]
- Introduced requirement for research protocol review by an independent committee [5]
- Stressed that considerations for the wellbeing of human subjects should always prevail over the interests of science and society [6]
- Addressed publication ethics and proper dissemination of research results [4]
The Belmont Report (1979)
Historical Context: Commissioned by the U.S. Congress in response to the Tuskegee Syphilis Study and other ethical scandals [4] [6]. The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research issued the report in 1979 [3] [7].
The Three Core Ethical Principles:
Respect for Persons: Individuals should be treated as autonomous agents, and persons with diminished autonomy (including children) are entitled to protection. This principle requires that subjects enter research voluntarily and with adequate information [4] [7].
Beneficence: Researchers must maximize possible benefits and minimize possible harms. This principle applies not only to individual investigators but to society at large, which must recognize that carefully considered research can yield important knowledge [4] [7].
Justice: Requires fair distribution of the benefits and burdens of research. The selection of subjects must be equitable and not target vulnerable populations (like institutionalized individuals or racial minorities) simply because of their availability [4] [7].
Technical Guide: Application to Pediatric Research
Regulatory Framework for Pediatric Research
The Belmont Report principles are embedded within the U.S. Code of Federal Regulations (45 CFR 46), with Subpart D providing additional protections for children involved in research [8] [4]. The following table outlines the risk categories that govern the approval of pediatric research:
Table: Institutional Review Board (IRB) Approval Categories for Pediatric Research
| Regulation Citation | Approval Criteria | Key Considerations |
|---|---|---|
| 45 CFR 46.404 [4] | Research not involving greater than minimal risk [8]. | "Minimal risk" is defined as the probability of harm or discomfort not greater than that encountered in daily life or routine examinations [8]. |
| 45 CFR 46.405 [4] | Research involving greater than minimal risk but presenting the prospect of direct benefit to individual subjects. | The risk must be justified by the anticipated benefit; the risk-benefit ratio must be at least as favorable as that presented by alternative approaches [9]. |
| 45 CFR 46.406 [4] | Research involving greater than minimal risk with no prospect of direct benefit, but likely to yield generalizable knowledge about the subject's disorder or condition. | The risk must represent a minor increase over minimal risk; the research experience must be reasonably commensurate with the subjects' actual medical, psychological, or social situations [8]. |
| 45 CFR 46.407 [4] | Research that does not fit the above categories but presents a reasonable opportunity to understand, prevent, or alleviate a serious problem affecting children's health or welfare. | Such research requires review by a national panel and approval by the Secretary of Health and Human Services [8]. |
Troubleshooting Common Ethical Challenges
FAQ 1: How do I properly obtain informed consent in pediatric research?
Issue: Confusion between consent, parental permission, and assent processes.
Resolution:
- Parental/Guardian Permission: Obtain informed consent from parents or legal guardians, as children cannot provide independent legal consent [8] [9]. This permission should include complete information about the study, including risks, benefits, and alternatives.
- Child Assent: Seek affirmative agreement from the child using age-appropriate language [8] [9]. The American Academy of Pediatrics recommends that children aged 7 years and older should be given the opportunity to assent, and their dissent should be respected unless the research offers direct therapeutic benefit not otherwise available [8].
- Adolescent Consent: For mature minors, provide a comprehensive study overview separately from parents, allowing them to ask questions freely. In certain cases (minimal risk research addressing issues that can only be studied in adolescents), a waiver of parental permission may be considered [8].
FAQ 2: How can we avoid coercion when recruiting children and their families?
Issue: Families may feel pressured to participate, especially when their primary physician is the researcher.
Resolution:
- Clear Communication: Explicitly state that refusal to participate or subsequent withdrawal will not affect the quality of clinical care [8]. Include this assurance in the consent form.
- Separation of Roles: Some literature suggests having research staff not directly involved in the child's clinical care approach the family initially to reduce perceived coercion [8].
- Appropriate Compensation: Compensate families for time and effort, but ensure payments are not so substantial that they become an undue influence that compromises voluntary participation [8].
FAQ 3: What strategies can minimize harm and discomfort in pediatric trials?
Issue: Research procedures can cause physical or psychological distress to children.
Resolution:
- Physical Discomfort: Use topical anesthetics before needle sticks, employ indwelling catheters to reduce repeated venipunctures, utilize micro-sampling techniques to minimize blood draw volumes, and establish maximum blood draw volumes based on scientific guidelines [9].
- Psychological Distress: Implement distraction strategies, ensure caregiver presence during procedures, use child-life specialists, design child-friendly settings, and explain procedures using age-appropriate language [9].
- Overall Burden: Design flexible protocols that minimize disruption to school and family routines [9].
FAQ 4: How do I determine if a child is capable of providing assent?
Issue: Uncertainty about assessing a child's capacity to understand research and provide meaningful assent.
Resolution:
- Developmental Considerations: The FDA and HHS regulations require IRBs to determine whether children are capable of providing assent based on age, maturity, and psychological state [8]. Capacity is generally considered as follows:
- Infants/Toddlers: Unable to provide assent; parental permission is mandatory [8].
- Young Children (approx. 7-11 years): Can understand basic concepts; simple explanation and affirmative agreement are required [8] [9].
- Adolescents (approx. 12-17 years): Can understand complex concepts similar to adults; should provide meaningful assent with detailed information [8].
The Scientist's Toolkit: Essential Frameworks for Ethical Pediatric Research
Table: Key Ethical Framework Solutions for Pediatric Researchers
| Tool or Framework | Primary Function | Application in Pediatric Research |
|---|---|---|
| Informed Consent Process [8] | Document voluntary participation with adequate information. | Requires dual approach: comprehensive parental permission and age-appropriate child assent. |
| Institutional Review Board (IRB) [8] | Independent review and approval of research protocols. | Specifically applies Subpart D of 45 CFR 46 to evaluate risk categories and adequacy of assent procedures for pediatric studies. |
| Data Safety Monitoring Board (DSMB) [9] | Independent ongoing safety review of trial data. | Provides additional oversight to protect vulnerable pediatric subjects, especially in high-risk or multi-site trials. |
| Risk-Benefit Assessment Framework [9] | Systematic evaluation of study justifiability. | Mandates that risks are minimized and reasonable in relation to potential benefits to the child or to knowledge about childhood conditions. |
| Age-Stratified Study Design [9] | Organizational approach to participant grouping. | Ensures appropriate representation and analysis of different developmental stages (neonates, infants, children, adolescents). |
Visualizing the Ethical Framework Evolution
Diagram: Evolution of Research Ethics Frameworks and Their Application to Pediatrics. This diagram traces the historical development of key ethical documents and illustrates how the core principles of the Belmont Report directly inform the specific applications and regulations that govern ethical pediatric research.
Understanding the historical context from the Nuremberg Code to the Belmont Report is essential for designing and conducting ethically sound pediatric research. The progression of these guidelines emphasizes that research involving children requires special protections, including appropriate parental permission, meaningful child assent, meticulous risk-benefit analysis, and equitable subject selection. By adhering to these foundational principles and implementing the practical troubleshooting strategies outlined in this guide, researchers can navigate complex ethical challenges while advancing medical knowledge to improve children's health outcomes.
Troubleshooting Guides
Guide 1: Addressing Common Challenges in the Assent Process
Problem: A 9-year-old child in a longitudinal study about cognitive development seems withdrawn and unwilling to participate in follow-up sessions, though their parent insists they continue.
- Potential Cause: The child may feel coerced by parental pressure or may not understand why continued participation is necessary. The initial assent process may not have adequately addressed the long-term nature of the study involvement.
- Solution:
- Private Conversation: Speak with the child alone, using age-appropriate language to re-explain the study and reaffirm that they can stop at any time without any trouble [10].
- Assess Understanding: Ask the child to explain back, in their own words, what they think the study is about and what they are being asked to do [10].
- Respect Dissent: Honor the child's non-verbal cues and reluctance. A child's deliberate objection (dissent) should be respected unless the research intervention offers a direct therapeutic benefit that is unavailable outside the research context [11] [12].
- Prevention: Implement ongoing assent processes in longitudinal studies, checking in with the child at each new stage or visit to ensure they remain a willing participant [13].
Problem: A researcher is preparing a clinical trial for a new pediatric asthma drug and is unsure how to categorize the risk level for Institutional Review Board (IRB) review.
- Potential Cause: The intervention involves a new chemical entity with unknown side effects in children, but it also holds the prospect of direct benefit for the participants whose asthma is not well-controlled by existing medications.
- Solution:
- Reference Regulatory Framework: Consult 45 CFR 46, Subpart D, which provides the categories of research involving children [8] [14].
- Categorize the Research: This study likely falls under 45 CFR 46.405, which covers research that presents more than minimal risk but offers the prospect of direct benefit to individual subjects [8].
- Document Justification: Prepare a rationale for the IRB explaining how the potential benefit justifies the risk, and detail the robust monitoring plans for participant safety [12].
- Prevention: Engage with the IRB during the protocol development phase to get preliminary feedback on the risk classification and consent/assent requirements.
Guide 2: Navigating Issues of Coercion and Undue Influence
Problem: A team is recruiting for a study on adolescent mental health and is concerned that a $100 compensation for a 2-hour survey might be coercive.
- Potential Cause: The incentive may be so large that it could unduly influence an adolescent's decision to participate, overpowering their consideration of the risks [8] [15].
- Solution:
- Reframe Compensation: Structure the payment as compensation for time and travel, rather than a reward for participation [8].
- Consult Guidelines: Refer to ethical guidelines that emphasize that incentives should not constitute undue influence [8] [15].
- IRB Review: Ensure the incentive plan is approved by the IRB, which will assess whether the payment is reasonable and not coercive for the specific population [14].
- Prevention: Pilot-test the consent and recruitment process with a small group from the target population to gauge their perception of the incentive.
Problem: A pediatrician-researcher is recruiting their own patients for a clinical trial. Parents are agreeing readily, but the researcher worries they may feel pressured.
- Potential Cause: Parents may fear that refusing to enroll their child could negatively affect the quality of their child's medical care or their relationship with the doctor [8].
- Solution:
- Clear Separation of Roles: Explicitly state during the consent process that the decision to participate or not will in no way affect the child's current or future medical care [8].
- Utilize Neutral Party: Where possible, have a member of the research team who is not directly involved in the child's clinical care approach the family for consent [8].
- Reinforce Voluntariness: The consent form and verbal explanation should strongly emphasize the voluntary nature of participation and the right to withdraw at any time [13] [14].
- Prevention: Implement clinic-wide protocols that clearly separate research activities from clinical care in the eyes of patients and families.
Frequently Asked Questions (FAQs)
Q1: At what age should we begin seeking a child's assent for research participation?
A: There is no universally fixed age. Federal regulations expect assent to be requested from children age 7 and older [8]. However, guidelines suggest this is a flexible standard. The American Academy of Pediatrics recommends that children as young as age 6 should be given the opportunity to decline participation [8] [14]. The key is to use a developmentally appropriate approach, tailoring the information and the process to the individual child's cognitive, emotional, and social maturity [12] [13].
Q2: What is the difference between "consent," "parental permission," and "assent"?
A:
- Consent: A legally valid authorization given by an autonomous adult (or an emancipated/minor with legal capacity) to participate in research. It is based on a full understanding of the research [11].
- Parental Permission: The permission granted by a child's parent or legal guardian, which is legally necessary for a child to participate in research. The ethical basis is to protect the child's welfare and interests, not to exercise parental autonomy [11] [12].
- Assent: The affirmative agreement of a child to participate. It is not legally binding but carries significant ethical weight. It involves providing the child with appropriate information and seeking their willing cooperation [11] [13].
Q3: Can we ever waive the requirement for parental permission?
A: Yes, but only under specific, justified circumstances. An IRB may waive parental permission if [12] [13] [14]:
- The research is designed to study conditions or behaviors for which parental knowledge could pose a risk to the child (e.g., studies on abuse, adolescent sexuality, or substance use).
- The research involves minimal risk.
- The waiver is essential to the practicability of the research.
- Additional protections are put in place, such as the appointment of an independent child advocate.
Q4: What should we do if a child assents but their parent refuses permission, or vice versa?
A: In most cases, both parental permission and the child's assent (when capable) are required. Therefore, if either party refuses, the child should not be enrolled [12]. The only potential exception is when the research intervention holds out a prospect of direct benefit that is unavailable outside the research and is considered the best medical option. In such rare cases, parental permission may override a child's dissent, but this requires careful ethical justification [11] [12].
Q5: How do we handle consent when a child participating in a long-term study reaches the legal age of adulthood?
A: When a participant reaches the legal age of majority during the study, researchers must obtain their own informed consent for continued participation [12]. This is a crucial step that respects the now-adult participant's autonomy and should be planned for in the study protocol.
Experimental Protocols & Methodologies
Protocol 1: Implementing a Developmentally-Staged Assent Process
Objective: To systematically obtain meaningful assent from child participants across a wide age range (6-17 years).
Methodology:
- Age 6-7: Provide a simple, brief oral description of what the child will do. Use concrete terms and visuals. Seek verbal assent. A witness should document the process [14].
- Age 8-13: Give a more complete oral explanation in layman's terms. Clearly state that participation is a choice. Continue to seek verbal assent with witness documentation [14].
- Age 14-17: Provide a written assent form written at an age-appropriate reading level. The process should mirror the adult consent process, and written assent should be obtained [14]. For mature adolescents, their agreement can be considered ethically equivalent to consent, though legally it may still be assent requiring parental permission [12].
Controls: The researcher's assessment of the child's comprehension is key. Staff should be trained to recognize and respond to non-verbal cues of dissent or anxiety [13] [10].
Protocol 2: Risk-Benefit Assessment for IRB Submission
Objective: To rigorously evaluate and document the risks and benefits of a proposed pediatric study to ensure it meets ethical and regulatory standards.
Methodology:
- Define "Minimal Risk": Establish that the probability and magnitude of harm or discomfort are not greater than those ordinarily encountered in daily life or during routine medical/psychological examinations [8] [12].
- Categorize the Research: Use the following framework from 45 CFR 46, Subpart D [8] [14]:
- 46.404: Research not involving greater than minimal risk.
- 46.405: Research involving greater than minimal risk but presenting the prospect of direct benefit to the individual subject.
- 46.406: Research involving greater than minimal risk and no prospect of direct benefit, but likely to yield generalizable knowledge about the subject's disorder or condition.
- 46.407: Research that cannot be approved under the previous categories but presents a reasonable opportunity to further understanding of a serious problem affecting children's health.
- Document the Analysis: For the IRB application, create a table that lists all procedures, distinguishes between therapeutic and non-therapeutic procedures, and categorizes the risk of each, justifying how the research fits into one of the above categories.
Controls: The final determination is made by the IRB. Engage in pre-submission consultation with the IRB for complex risk profiles.
Data Presentation
Table 1: Categorization of Pediatric Research Risk According to 45 CFR 46 Subpart D
| IRB Approval Category | Risk Level | Potential for Direct Benefit | Key Justifying Condition | Example of Applicable Research |
|---|---|---|---|---|
| 46.404 | No greater than minimal risk | Not required | Adequate provisions are made for solicting assent and permission. | Behavioral surveys on non-sensitive topics; analysis of leftover blood samples. |
| 46.405 | Greater than minimal risk | Yes, for the participant | Risk is justified by the anticipated benefit to the subject; the risk-benefit ratio is at least as favorable as alternatives. | Clinical trial of a new chemotherapeutic agent for pediatric cancer. |
| 46.406 | Greater than minimal risk, minor increase over minimal | No | The research is likely to yield vital knowledge about the participant's condition; procedures are commensurate with the participants' actual experiences. | A lumbar puncture to study CNS leukemia in children with the disease. |
| 46.407 | Not allowable under 404-406 | Not necessarily | Research presents a reasonable opportunity to understand a serious health problem; the study is ethically sound and will be conducted with adequate safeguards. | A study that is vital for public health but involves a procedure with risk that cannot be minimized to fit categories 405 or 406. |
Table 2: Guidelines for Child Assent by Developmental Stage
| Age Group | Cognitive Capacity | Recommended Assent Process | Documentation | Special Considerations |
|---|---|---|---|---|
| < 6 years | Limited understanding; cannot provide meaningful assent. | N/A (Focus on parental permission) | N/A | Observe for distress (dissent) and comfort accordingly. Respect non-deliberate objections (crying, withdrawal) where possible [11]. |
| 6-7 years | Concrete thinking; short attention span. | Simple verbal explanation. Use pictures/toys. Seek verbal agreement. | Signature of a witness on consent form. | Use short sentences and simple concepts. The line between assent and dissent is subtle [14]. |
| 8-13 years | Developing logical reasoning; can understand cause and effect. | More detailed verbal explanation in lay terms. Explain voluntary nature and right to withdraw. | Signature of a witness on consent form. | They can understand the purpose of research. Begin to use written materials to supplement verbal explanation [14]. |
| 14-17 years | Abstract thinking; capable of understanding complex concepts and consequences. | Formal, written assent document, tailored to reading level. Process should mirror adult consent. | Signed written assent form. | For mature adolescents, their agreement carries significant weight. Consider their input in decision-making seriously [12] [14]. |
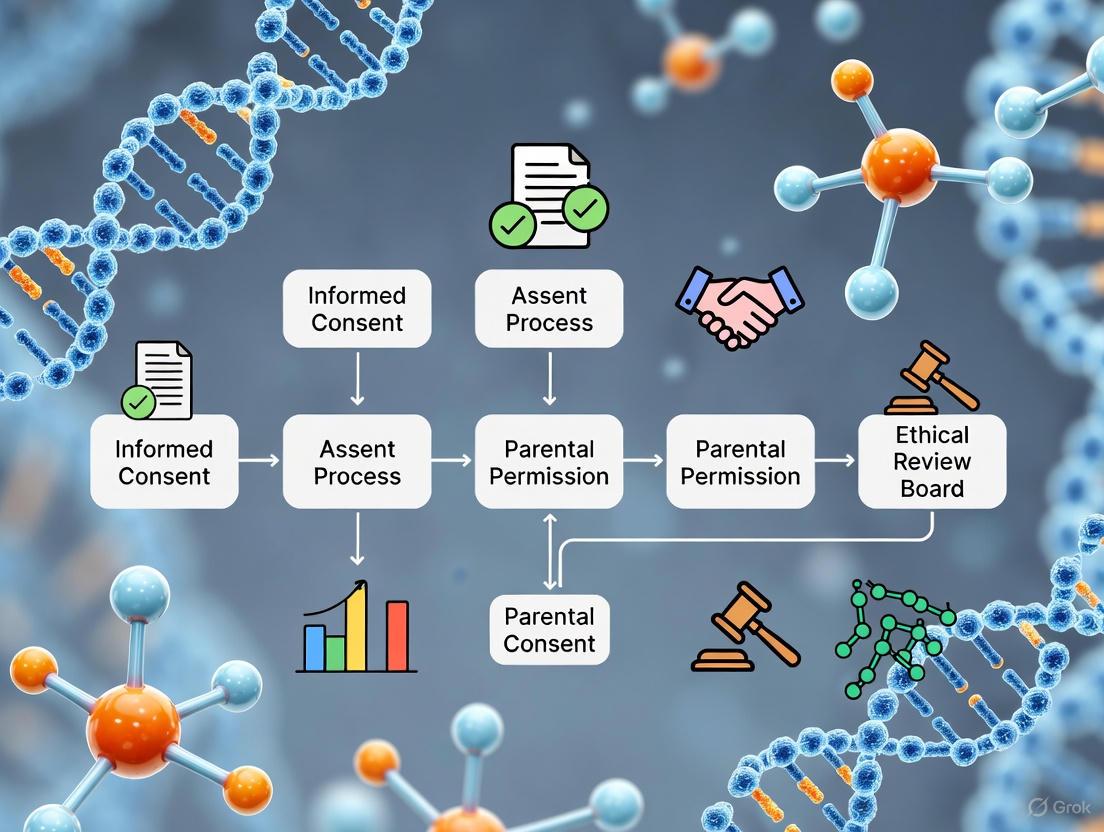
Visualizations: Ethical Pathway for Pediatric Research Approval
DOT Language Script for Diagram
Research Participant Enrollment Decision Tree
This diagram illustrates the logical sequence of ethical and regulatory checks required before a child can be enrolled in a research study, integrating the principles of Respect for Persons (through assent/parental permission), Beneficence (through risk categorization), and Justice (through fair application of procedures).
The Scientist's Toolkit: Essential Materials for Ethical Pediatric Research
| Item/Reagent | Function & Ethical Justification |
|---|---|
| IRB-Approved Protocol | The foundational document ensuring the study design is scientifically sound and ethically defensible. It is the primary tool for protecting participant welfare [8] [14]. |
| Parental Permission Form | A document written at an 8th-grade reading level or lower to ensure comprehensibility. It legally authorizes the child's participation and embodies the principle of Respect for Persons for the family unit [8] [14]. |
| Developmentally-Staged Assent Scripts & Forms | Age-appropriate tools (verbal scripts for young children, written forms for adolescents) to engage the child in the decision-making process, respecting their developing autonomy [13] [14] [10]. |
| Certificate of Confidentiality | A critical protective document issued by a government agency to shield sensitive research data (e.g., on illegal behaviors) from subpoena, thereby minimizing legal risks to participants and families [14]. |
| Resource Referral List | A list of counseling, medical, or social services to provide to participants and families if the research raises psychosocial stress or reveals unmet needs, fulfilling the principle of Beneficence beyond the immediate research scope [14] [10]. |
Troubleshooting Guide: Common Scenarios in Pediatric Research
Scenario 1: Determining the Correct Risk Category for Your Study
- Problem: A researcher is unsure whether their study on a new asthma monitoring device for children presents "minimal risk" or a "minor increase over minimal risk."
- Solution:
- Step 1: Define the "minimal risk" baseline. "Minimal risk" means the probability and magnitude of harm or discomfort anticipated in the research are not greater than those ordinarily encountered in daily life or during routine physical or psychological examinations [16]. Examples include obtaining small blood samples, urinalysis, or non-invasive swabbing [16].
- Step 2: Compare your procedures to this baseline. For the asthma device, consider the invasiveness, time commitment, and potential discomfort against a healthy child's daily life or routine check-ups.
- Step 3: Consult the IRB early. The IRB will make the final determination based on the specific procedures and the subject population (e.g., a procedure deemed minimal risk for healthy children may not be for children with a specific condition) [16].
Scenario 2: Navigating Parental Permission Requirements
- Problem: A research team is recruiting for a study that fits the §46.406 category (greater than minimal risk, no direct benefit). They are unsure if they need permission from one or both parents.
- Solution:
- Step 1: Identify your study's approved category. The regulatory requirements for parental permission are directly tied to the category of research, as summarized in the table below.
- Step 2: Determine "reasonable availability." For categories requiring permission from both parents, you must obtain it from both, unless one is "not reasonably available" [16]. A parent may be considered not reasonably available if they are unknown, deceased, incompetent, incarcerated, or not reasonably contactable. Detailed documentation of attempts to contact is crucial.
- Step 3: Verify legal authority. Ensure the person providing permission is a biological parent, adoptive parent, or has a court order granting them the legal authority to consent to medical care for the child [16].
Scenario 3: Waiving Assent for a Complex Clinical Trial
- Problem: An investigator proposes to waive the requirement for child assent in a cancer trial, arguing the young participants (ages 10-15) cannot understand the complex nature of the disease and treatment.
- Solution:
- Step 1: The IRB must evaluate this request carefully. The regulations require the IRB to determine that adequate provisions are made for soliciting assent, unless the capability of some or all of the children is so limited that they cannot reasonably be consulted [16].
- Step 2: Consider age, maturity, and psychological state. A blanket waiver for all children in this age range is unlikely to be justified. A more appropriate approach would be to develop an age-appropriate assent process and allow for a case-by-case waiver only for children who demonstrably lack the capacity to assent.
- Step 3: Document the rationale. Any decision to waive the assent requirement must be well-documented by the IRB, including the justification for why the children's capabilities are so limited that they cannot be consulted.
Frequently Asked Questions (FAQs)
Q1: What are the core ethical principles underpinning Subpart D and FDA regulations for children? The regulations are built upon foundational ethical principles outlined in the Belmont Report: Respect for Persons, Beneficence, and Justice [4]. Respect for Persons requires seeking parental permission and, when appropriate, the child's assent. Beneficence obligates researchers to maximize benefits and minimize harms. Justice ensures the fair distribution of the benefits and burdens of research, protecting vulnerable populations like children from bearing a disproportionate share of research risks [4].
Q2: What is the key regulatory difference between 45 CFR 46 Subpart D and FDA regulations (21 CFR 50 Subpart D)? The primary difference is the scope of authority. 45 CFR 46 Subpart D applies to all research conducted or supported by the Department of Health and Human Services (HHS) [17]. The FDA's regulations (21 CFR 50 Subpart D), however, apply to all clinical investigations involving FDA-regulated products (drugs, biologics, devices, etc.), regardless of funding source [18]. The substantive protections for children are harmonized between the two sets of regulations.
Q3: My study involves a minor increase over minimal risk with no direct benefit to the child. Is it automatically disapproved? No, not automatically. Such research may be approved under §46.406 if it meets three strict criteria:
- The risk represents only a minor increase over minimal risk.
- The interventions are reasonably commensurate with the child's actual or expected medical, dental, psychological, social, or educational situations.
- The research is likely to yield generalizable knowledge about the child's disorder or condition that is of vital importance for understanding or ameliorating it [16].
Q4: Who is considered a "parent" for providing permission? A "parent" is defined as a child's biological or adoptive parent. A legal guardian may also provide permission but must provide documentation of their legal authority to consent for the child's general medical care. It is critical to distinguish between a caregiver who has physical custody and a person with legal decision-making authority [16].
Q5: What recent regulatory changes should I be aware of? In October 2024, HHS issued a final rule updating subparts B, C, and D of 45 CFR part 46. For subpart D, these changes were primarily technical, updating citations to align with the 2018 revisions to the Common Rule ("the 2018 Requirements") and correcting a technical error [17]. This update ensures consistency but does not introduce substantive changes to the protections for children. Researchers should confirm with their IRB which set of requirements (pre-2018 or 2018) their study falls under [17].
Regulatory Reference Tables
Table 1: Categories of Pediatric Research Permissible under 45 CFR 46 Subpart D and 21 CFR 50 Subpart D
| Category & Citation | Risk/Benefit Profile | Key Criteria for Approval | Parental Permission Requirement |
|---|---|---|---|
| §46.404 / §50.51 [16] | Research not involving greater than minimal risk. | Procedures must not exceed minimal risk thresholds. | One or both parents, as determined by the IRB. |
| §46.405 / §50.52 [16] | Research involving greater than minimal risk but presenting the prospect of direct benefit to the subject. | 1. Risk justified by anticipated benefit.2. Risk-benefit ratio at least as favorable as alternatives. | One or both parents, as determined by the IRB. |
| §46.406 / §50.53 [16] | Research involving greater than minimal risk and no prospect of direct benefit, but likely to yield generalizable knowledge about the subject's disorder/condition. | 1. Risk is a minor increase over minimal.2. Experiences are reasonably commensurate with the subject's situation.3. Knowledge is of vital importance. | Permission of both parents is required, unless one is deceased, unknown, incompetent, or not reasonably available. |
| §46.407 / §50.54 [16] | Research not otherwise approvable that addresses a serious problem affecting children's health or welfare. | 1. Presents a reasonable opportunity to understand, prevent, or alleviate a serious problem.2. Conducted per sound ethical principles.3. Adequate provisions for assent and permission. | Permission of both parents is required, unless one is deceased, unknown, incompetent, or not reasonably available. |
Table 2: The Researcher's Regulatory Toolkit
| Item | Function in Pediatric Research | Relevant Source |
|---|---|---|
| Institutional Review Board (IRB) | Reviews, approves, and monitors research to ensure ethical and regulatory compliance. Must include members knowledgeable about working with children [4] [18]. | 21 CFR 56, 45 CFR 46.107 |
| Parental Permission Form | Documents the informed agreement of the parent(s) or guardian(s) for the child to participate. Must contain all required elements of informed consent, presented in a comprehensible manner. | 21 CFR 50, 45 CFR 46.116 |
| Child Assent Process & Document | Affirmative agreement from the child to participate. The process and documentation must be appropriate to the child's age, maturity, and psychological state [16]. | 45 CFR 46.408, IRB Policies |
| Protocol-Specific Risk Assessment | A detailed justification classifying the study into one of the four regulatory categories, justifying the level of risk and the risk-benefit relationship. | 45 CFR 46.404-407 |
| Documentation of Legal Guardianship | Court orders or other legal documents verifying an individual's authority to provide permission for a child's participation in research [16]. | State Law, IRB Policies |
Experimental Protocol: Navigating the IRB Approval Pathway for Pediatric Research
This protocol outlines a systematic methodology for securing IRB approval for research involving child participants, within the ethical framework of pediatric consent challenges.
1. Pre-Submission Analysis
- Objective: To internally classify the study and prepare all necessary justifications before formal IRB submission.
- Methodology:
- Risk Categorization: Use the tables above to conduct a preliminary classification of your study (§46.404, .405, .406, or .407). Justify this classification in writing, citing specific study procedures and the "minimal risk" baseline [16].
- Benefit Analysis: Clearly delineate and document any prospect of direct benefit to the participant. If there is no direct benefit, articulate the vital importance of the generalizable knowledge to be gained about the children's disorder or condition [16].
- Permission & Assent Strategy: Based on the proposed category, determine the required number of parents for permission. Design an age-appropriate assent process and document, including the language to be used and any visual aids [16].
2. IRB Submission and Justification
- Objective: To present a complete and defensible application to the IRB.
- Methodology:
- Complete the IRB Application: Fill out all relevant sections, explicitly stating the proposed category of research.
- Upload Supporting Documents: Include the finalized study protocol, the parental permission form, child assent documents, and the internal justification memo from Step 1.
- Justify Vulnerable Population: Explain why children are the necessary population for the research and detail the additional safeguards incorporated to protect their welfare [18].
3. Post-Review Action
- Objective: To efficiently address any IRB stipulations and implement the approved protocol.
- Methodology:
- Respond to Stipulations: If the IRB disagrees with the initial category (e.g., determines the risk is higher than proposed), revise the application and justifications accordingly.
- §46.407 Review: If the IRB believes the study may only be approvable under §46.407, cooperate with the IRB and any convened panel of experts to review the study's merits [16].
- Implement Approved Process: Train all study staff on the final, approved procedures for obtaining and documenting parental permission and child assent.
Regulatory Pathway Visualization
The following diagram illustrates the logical decision pathway for categorizing pediatric research under Subpart D, which is crucial for troubleshooting IRB submission issues.
Core Definitions and Regulatory Framework
What are the foundational definitions of "Minimal Risk" and "Greater than Minimal Risk"?
The US Code of Federal Regulations (CFR) provides the specific definitions that govern human subjects research. These definitions are critical for determining the level of review required by an Institutional Review Board (IRB) [4] [19].
Minimal Risk is definitized as a scenario where "the probability and magnitude of harm or discomfort anticipated in the research are not greater in and of themselves than those ordinarily encountered in daily life or during the performance of routine physical or psychological examinations or tests" [19] [20] [21].
Greater than Minimal Risk is any research where the probability and magnitude of harm or discomfort are greater than this encountered daily life standard [19].
Table: Foundational Definitions of Research Risk
| Risk Category | Regulatory Definition | Key Interpretation |
|---|---|---|
| Minimal Risk | "The probability and magnitude of harm or discomfort... are not greater... than those ordinarily encountered in daily life or during... routine... examinations or tests." [19] [20] | Risks are comparable to those of a healthy person's routine clinical visits or daily activities [20]. |
| Greater than Minimal Risk | Anything determined to be above the "minimal risk" threshold. [19] | The risks of the research exceed those encountered in daily life or routine exams. |
How do these risk categories impact IRB review procedures?
The initial risk determination, made prior to research initiation, directly dictates the type of IRB review a study must undergo [19].
- Greater than Minimal Risk Studies: Must receive full committee review at a convened IRB meeting [19].
- Minimal Risk Studies: May be eligible for expedited review or, in some cases, exempt review, which generally involves a faster process conducted by the IRB chair or a designated reviewer rather than the full board [19] [20].
Risk Categorization in Pediatric Research
What special protections exist for children in research?
Children are classified as a vulnerable population, and the federal regulations in 45 CFR 46, Subpart D provide additional protections for them [4] [19]. The ethical framework for pediatric research is built upon three core principles from The Belmont Report [4]:
- Respect for Persons: Recognizing the autonomy of individuals and requiring informed consent; those with diminished autonomy (like children) are entitled to special protection.
- Beneficence: The obligation to maximize possible benefits and minimize possible harms.
- Justice: Ensuring the fair distribution of the benefits and burdens of research [4].
For pediatric research that does not offer a prospect of direct benefit to the child, the regulations are particularly strict. Such non-beneficial research is generally permissible only when it presents no more than minimal risk [21]. A special category, "a minor increase over minimal risk," exists for certain non-beneficial research in children with a condition or disorder, but this category is not explicitly defined in the regulations and requires careful ethical deliberation [21].
What are examples of procedures typically considered "Minimal Risk" vs. "Greater than Minimal Risk"?
The following table provides concrete examples to help illustrate the distinction between these risk categories, with a focus on pediatric research contexts.
Table: Examples of Procedures by Risk Category in Pediatric Research
| Minimal Risk Procedures | Greater than Minimal Risk Procedures |
|---|---|
| Blood sampling from a vein (within volume limits based on total blood volume for children) [20] | Administration of virtually any investigational drug or biologic, even if approved for other uses and known to have a mild safety profile [20] |
| MRI without contrast and without sedation [20] | MRI with contrast and/or sedation [20] |
| "Surface" (external only) imaging, without contrast agents [20] | A clinical investigation of a device that involves invasive sampling [20] |
| Placement of a peripheral venous line for less than 24 hours [20] | Research involving deceptive procedures that participants would find offensive if not prospectively agreed to [19] |
| Most research on regular and special education instructional strategies [19] | Surveys or interviews of children where disclosure of responses could harm the subject's reputation or employability [19] |
FAQs and Troubleshooting for Researchers
FAQ: How is "daily life" defined for minimal risk determinations?
This is a primary source of ongoing debate and ambiguity among research ethicists [21]. Several interpretations exist, and there is no universal consensus:
- The "Average, Normal, Healthy" Child Standard: Refers to the risks faced by average, normal, healthy children in their daily lives [21].
- The "Relative" Interpretation: Refers to the risks that the specific child being enrolled in research faces in their own daily life. This interpretation is criticized by some as potentially unjust, as it could allow disadvantaged children to be exposed to higher levels of risk in research [21].
- The "Routine Examinations" Interpretation: Some ethicists argue that the "daily life" clause is problematic and should be eliminated, using only the "routine physical or psychological examinations" as the standard [21].
Troubleshooting Tip: When designing your study, consult with your IRB early to understand which interpretation they apply. Justifying your risk assessment using the "average, normal, healthy" child standard is often a prudent starting point.
FAQ: Our study involves an approved drug. Is this automatically minimal risk?
No. According to FDA guidance, the administration of virtually any investigational drug or biologic is considered greater than minimal risk, even if the product is approved for the indication being studied or for other indications and is known to have a generally mild safety profile [20]. The determination is based on the regulatory definition of risk, not just the clinical profile of the product.
FAQ: What is a "minor increase over minimal risk" in pediatric research?
This is a derivative category specific to pediatric research regulations. It applies to non-beneficial research for children with a condition or disorder [21]. While not explicitly defined, it is intended to allow for a small, justifiable escalation in risk beyond the minimal risk threshold for studies that are vital to understanding the child's condition. The ethical rationale is not fully detailed in the regulations, and such proposals often require robust justification to the IRB [21].
Experimental Protocol: Risk Categorization Workflow
The following diagram outlines the logical decision process for categorizing study risk, a key methodological step in protocol development.
Research Reagent Solutions: Ethical and Regulatory Frameworks
For researchers in pediatric drug development, the following "reagents" are essential for constructing an ethically sound study.
Table: Essential Frameworks for Pediatric Research Ethics
| Framework/Reagent | Function in Research Protocol |
|---|---|
| The Belmont Report [4] | Provides the foundational ethical principles (Respect for Persons, Beneficence, Justice) that justify and constrain research involving human subjects. |
| 45 CFR 46, Subpart D [4] [21] | The specific regulatory "tool" that outlines the conditions under which pediatric research is permissible, including the risk categories of §46.404, §46.405, etc. |
| Declaration of Helsinki [4] | A foundational international policy statement on ethical principles for medical research involving human subjects, including special provisions for vulnerable groups. |
| Institutional Review Board (IRB) | The formal committee that operates as the final "validation assay," reviewing, approving, and monitoring research to protect the rights and welfare of human subjects. |
The dual permission model, requiring both parental consent and child assent, represents a cornerstone of ethical pediatric research. This framework is designed to protect children's rights and well-being while acknowledging their developing autonomy. Parental consent provides legal authorization for a child's participation, grounded in the parent's responsibility to safeguard their child's welfare. In contrast, child assent represents the child's affirmative agreement to participate, reflecting respect for their emerging capacity to understand and engage with the research process. Despite its ethical importance, implementing this model presents complex challenges for researchers, including determining when children are capable of providing meaningful assent, adapting processes for diverse cultural and socioeconomic settings, and managing situations where parents and children disagree. This technical support guide addresses these challenges through practical troubleshooting advice, evidence-based methodologies, and clear protocols to ensure ethically sound and regulatory-compliant research practices.
FAQs: Core Concepts and Regulatory Framework
1. What is the fundamental difference between parental consent and child assent?
Parental consent is the legal permission granted by a parent or guardian, representing a decision made in the best interest of the child. Child assent is the child's affirmative agreement to participate, demonstrating respect for the child as a person with developing autonomy. Federal regulations require both for research involving greater than minimal risk with no direct benefit, unless one parent is deceased, unknown, incompetent, not reasonably available, or when only one parent has legal responsibility [22]. The table below summarizes the key distinctions:
Table: Distinguishing Parental Consent from Child Assent
| Feature | Parental Consent | Child Assent |
|---|---|---|
| Legal Status | Legally authoritative permission | Ethical affirmation without legal authority |
| Primary Basis | Best interest of the child, substituted judgment | Understanding and willingness to participate |
| Regulatory Trigger | Required for all pediatric research | Required when children are capable of providing it (IRB judgment) |
| Documentation | Formal signed consent form | Varies (signed form, verbal agreement, documented dialogue) |
2. When is a child considered capable of providing assent?
A child's capability to assent depends on age, maturity, and psychological state, not a fixed age threshold. Competence is judged by the child's ability to understand and retain relevant information, weigh this information in making a mature judgment, come to a decision, and communicate that decision [23]. The level of information provided should be commensurate with the child's development. For older children and teenagers, this may approach the complexity of adult consent, while for younger children, it may involve explaining basic procedures and their feelings about participation.
3. What should researchers do if a parent consents but the child dissents?
A child's dissent should be taken seriously. Ethically, researchers should respect the child's refusal in most non-therapeutic research contexts. For research offering direct therapeutic benefit, the balance may shift, but the child's concerns must be addressed thoroughly. The process should involve exploring the reasons for dissent, providing additional information at the child's level of comprehension, and ensuring the child does not feel coerced. Overriding a persistent dissent risks violating the ethical principle of respect for persons and can cause significant distress to the child participant.
4. How is "not reasonably available" defined for dual parental permission?
Regulations permit single-parent consent when one parent is not "reasonably available." Practical scenarios from research include fathers who are deployed with the military, incarcerated, living out of state, or not involved in the mother's life [22]. Consultations with IRB officials and legal counsel have produced criteria for this determination, emphasizing factors like the father's presence on the birth certificate and the feasibility of contact. An algorithm can guide this decision, starting with establishing the father's legal status and then assessing practical availability [22].
Troubleshooting Guide: Common Scenarios and Solutions
Scenario 1: Determining Child's Capacity to Assent in Low-Resource Settings
- Challenge: In low-income settings, factors like low education levels, high illiteracy, and different cultural norms around childhood autonomy complicate assessments of a child's capacity to understand research [23].
- Recommended Protocol:
- Contextual Assessment: Evaluate the child's decision-making capacity against the complexity of decisions they are accustomed to making in daily life, using local rather than Western standards [23].
- Simplify Communication: Use verbal explanations, visual aids, and participatory methods like role-playing instead of relying on written assent forms, which test literacy more than understanding.
- Engage Community: Consult with parents, teachers, and community leaders to understand local perceptions of childhood competence and to develop culturally appropriate assent materials.
Scenario 2: Managing Disagreement Between Parent and Child
- Challenge: A parent provides consent, but the child, particularly an adolescent, expresses clear dissent.
- Recommended Protocol:
- Facilitate Dialogue: Create a safe space for the family to discuss the research, the child's concerns, and the parent's perspective.
- Explore Motivations: Gently explore the reasons behind the child's dissent (e.g., fear of a procedure, misunderstanding, time commitment).
- Prioritize the Child's Will: In research that does not offer a direct therapeutic benefit to the child, the child's dissent should typically be honored. Document the process thoroughly.
Scenario 3: Applying Dual Parental Consent with Complex Family Structures
- Challenge: Navigating consent requirements with unmarried parents, same-sex couples, or when a legal guardian is not a biological parent.
- Recommended Protocol:
- Establish Legal Authority: First, determine who has legal decision-making authority for the child. The name(s) on the birth certificate is a primary indicator, though state laws vary [22].
- Consult IRB and Legal Counsel: For complex cases (e.g., a mother's legal spouse is not the biological father but is on the birth certificate), seek guidance. The consensus is that the legal parent on the birth certificate must provide permission [22].
- Document Rationale: Meticulously document the family structure, the determination of who provided consent, and the rationale for why another individual was deemed not to have legal authority or was "not reasonably available."
The following workflow diagram outlines the key decision points and processes in the dual permission model, integrating both parental consent and child assent procedures.
Experimental Protocols and Methodologies
Protocol 1: Implementing a Tiered Assent Process
A tiered assent process recognizes that children are not a homogeneous group and tailors the assent process to the individual child's developmental level.
- Materials: Age-appropriate information sheets (using pictures and simple language for young children; more detailed documents for adolescents), assent forms or verbal script templates, and a documentation log.
- Procedure:
- Pre-Assessment: The researcher, in consultation with parents, makes an initial judgment of the child's capacity level (e.g., early childhood, school-age, adolescent).
- Information Disclosure: The researcher provides a developmentally appropriate explanation of the research. For a young child: "We are doing a study to help kids. This will involve a special check-up and a game. It might be a little boring, but it won't hurt." For an adolescent, provide information similar to an adult consent form but in plain language.
- Assessment of Understanding: Ask open-ended questions to gauge comprehension (e.g., "Can you tell me in your own words what we are going to do?").
- Soliciting Agreement: Ask for the child's agreement to participate. For younger children, a verbal "yes" is sufficient. For older children, a signed simplified form can be used.
- Documentation: Document the process, the information provided, the child's response, and the researcher's assessment of the child's understanding.
Protocol 2: Algorithm for Determining "Reasonably Available" Second Parent
This protocol, derived from a large-scale newborn screening study, helps standardize decisions when dual parental consent is required but one parent is absent [22].
- Materials: IRB-approved guidelines, legal counsel consultation notes, screening questionnaire for mothers.
- Procedure:
- Establish Legal Status: If the mother has consented but the father is absent, determine if the father is listed on the birth certificate. If not listed and not involved, he may be considered not reasonably available.
- Inquire About Availability: If the father is on the birth certificate, ask the mother if the father will be coming to the hospital or is otherwise available (e.g., by phone).
- Attempt Contact: If the father is available by phone, attempt to contact him to obtain consent.
- Final Determination: If the father is out of state and the mother will not provide contact information, or if he is unavailable due to military deployment, incarceration, or other definitive reasons, document the rationale for deeming him "not reasonably available" and proceed with the mother's consent alone [22].
Table: Quantitative Data on Father Availability in a Newborn Screening Study
| Scenario | Number of Cases | Percentage of Total (%) |
|---|---|---|
| Total Families Approached | 3001 | 100% |
| Fathers Not Present | 589 | 19.6% |
| Fathers Deemed Not Reasonably Available | 158 | 5.3% |
| Primary Reasons: Military, Incarceration, Out-of-State, Not Involved | (Not specified) | (Not specified) |
| Enrollment when Both Parents Available & Consented | (Not specified) | 64% |
Table: Key Research Reagent Solutions for Ethical Consent and Assent
| Tool / Material | Primary Function | Application Notes |
|---|---|---|
| Developmentally-Structured Assent Forms | Provides a standardized yet flexible template for soliciting and documenting child assent. | Create multiple versions tailored to age groups (e.g., 6-9, 10-14, 15-17). Use large font, visuals, and simple checkboxes for younger children. |
| Visual Aids & Storyboards | Enhances comprehension for children with low literacy or in multilingual contexts. | Use pictures or simple diagrams to explain research procedures, blood draws, or clinic visits. |
| IRB-Approved Decision Algorithm | Guides consistent application of regulatory requirements for dual parental consent. | A flow chart for determining when a parent is "not reasonably available," vetted by the IRB and legal counsel [22]. |
| Cultural Consultation Framework | Aids in adapting consent/assent materials and processes for specific cultural or local contexts. | Involves engaging community advisors to review materials for cultural appropriateness and conceptual understanding of research. |
| Documentation and Audit Log | Ensures a verifiable record of the entire dual permission process for regulatory compliance. | Templates for recording the consent/assent discussion, questions asked, answers given, and the final outcome. |
From Theory to Practice: Implementing Effective Consent and Assent Procedures
Structuring the Informed Consent Form for Parental Comprehension
For researchers and drug development professionals, obtaining valid informed consent in pediatric studies presents unique ethical and practical challenges. The process requires more than just translating an adult consent form; it demands a deliberate restructuring to ensure parental comprehension and respect for the developing autonomy of the child. This technical support center addresses the specific hurdles you may encounter when designing these critical documents, providing troubleshooting guides and detailed protocols to enhance clarity, uphold ethical standards, and support the validity of your research data.
FAQs on Parental Comprehension in Informed Consent
1. What reading level should a parental consent form target, and how can I achieve it? Parental consent forms should be written at or below an 8th-grade reading level to ensure broad comprehension [24] [25] [26]. This is a best practice endorsed by multiple research institutions.
- Troubleshooting Tip: Avoid technical jargon and use everyday language. For instance, replace "The parturient will undergo phlebotomy" with "We will take a small amount of blood from you" [24] [27].
- Methodology: Use tools like the Flesch-Kincaid Grade Level test in word processors to check readability [26]. Before submission, have a colleague or a non-specialist read the document to identify confusing passages.
2. How is "Key Information" different in a parental consent form? The 2018 Common Rule requires consent forms to begin with a "concise and focused" presentation of key information [27] [25]. For parents, this means immediately addressing what is most relevant to their decision about their child's participation.
- Key Information to Include [27] [25]:
- A clear statement that the study is research and participation is voluntary.
- A summary of the research purpose, what the child will do, and the study's duration.
- The most significant risks or discomforts the child might experience.
- Any potential benefits for the child or others.
- Alternatives to participation, if any.
- Troubleshooting Tip: This section should be a summary, not an exhaustive list. It should balance reasons to participate (e.g., contributing to knowledge) with reasons not to (e.g., significant time commitment or known risks) [27].
3. When can I request a waiver of signed parental consent? An IRB may grant a waiver of documentation (signature) for parental consent in specific situations. The following table outlines the primary regulatory scenarios [24] [14]:
| Waiver Scenario | Regulatory Conditions | Common Application in Pediatric Research |
|---|---|---|
| Potential Harm from Breach of Confidentiality | The consent form is the only link between the child and the research; the principal risk is a confidentiality breach [24]. | Research on sensitive topics (e.g., adolescent substance use, mental health) where storing a signed form poses the primary risk to the participant [14]. |
| Minimal Risk Research | The research presents no more than minimal risk and involves no procedures requiring written consent outside the research context [24]. | Minimal risk surveys, educational tests, or observational research where a signed form is not standard practice [24]. |
| Cultural Norms or Impracticality | Signing forms is not the norm in the participants' cultural community, and the research is minimal risk [24]. | Research within distinct cultural groups or communities where a signed form is not customary. The IRB may require an alternative documentation mechanism [24]. |
4. How do I adapt the consent process for a child's age? The consent process involves two parts: parental permission and child assent. Assent is the child's affirmative agreement to participate. The method for obtaining assent should be appropriate to the child's age, literacy, and maturity level [14] [28].
| Age Range | Recommended Assent Procedure | Documentation |
|---|---|---|
| Ages 6-7 | Provide a simple oral description of what the child will do. | Request verbal agreement. Document via a witness signature on the consent form [14]. |
| Ages 8-12 | Give a more complete oral explanation using lay terminology. | Request verbal assent. Document via a witness signature [14]. |
| Ages 13-17 | Use a written assent form written at an appropriate reading level (e.g., 5th-7th grade) [28]. | Obtain the child's signature on a dedicated, age-appropriate assent document [14]. |
5. What are the common formatting mistakes that hinder parental comprehension?
- Mistake: Using small fonts and dense paragraphs.
- Solution: Use a 12-point font or larger. Break up text with bullet points, ample white space, and headers. For some populations, such as the elderly or visually impaired, a larger type size may be appropriate [24].
- Mistake: Writing in the first person ("I understand...").
- Solution: Write directly to the parent in the second person ("You") [24] [26]. Use active voice (e.g., "We will send you a summary of the results") instead of passive voice (e.g., "A summary of results will be sent...") [26].
Experimental Protocols for Consent Form Development
Protocol 1: Readability and Comprehension Testing
This methodology ensures your consent form is understandable to the target parent population.
- Step 1 – Drafting: Use an institutional template [25] [28] to ensure all regulatory elements are included.
- Step 2 – Readability Analysis: Run the draft through the Flesch-Kincaid tool in your word processor. Aim for a score at or below an 8th-grade level [26].
- Step 3 – Plain Language Review: Replace or define all technical terms using resources like the Stanford University Glossary of Lay Terms or the University of Michigan Plain Language Medical Dictionary [27].
- Step 4 – Peer Feedback: Have a colleague unfamiliar with the study review the form for clarity and logic.
- Step 5 – Pilot Testing (if possible): Administer the form to a small group of individuals similar to your target population and test their understanding of key concepts.
Protocol 2: Designing a Multi-Stage Consent and Assent Process
This protocol outlines the workflow for ethically enrolling a pediatric participant, integrating both parental permission and child assent.
The following diagram visualizes the sequential workflow and logical relationships in the pediatric consent and assent process:
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key resources for developing and managing ethically sound informed consent processes in pediatric research.
| Tool / Resource | Function | Source / Example |
|---|---|---|
| IRB Consent Template | Standardized document ensuring all regulatory elements of informed consent are included. | Institution-specific templates (e.g., UCI IRB, University of Michigan templates) [24] [25]. |
| Glossary of Lay Terminology | Replaces complex medical/scientific terms with simple, understandable language. | Stanford University Glossary of Lay Terms; University of Michigan plain language medical dictionary [27]. |
| Readability Testing Software | Objectively measures the grade level of consent documents to ensure appropriateness. | Flesch-Kincaid Grade Level test (built into many word processors) [26]. |
| Informed Consent Library (ICLibrary) | An internal system for study staff to access the currently approved and master versions of informed consents. | Example: Boston Children's Hospital ICLibrary [27]. |
| Short Form Consent Documents | Translated abbreviated documents used with non-English speaking participants when the full consent is presented orally. | Available in multiple languages (e.g., Spanish, Chinese, Arabic) [27]. |
Developing Age-Appropriate Assent Processes for Children
FAQs: Navigating Common Ethical Challenges
FAQ 1: At what age should I begin obtaining assent from child participants?
While federal regulations typically require formal assent for children aged 7 and older, ethical best practices support involving children in the decision-making process at much younger ages [29] [30]. Even infants and toddlers can non-verbally express preference and aversion. The key is to tailor the process to the child's developmental capacity rather than relying solely on legal minimums [29] [31].
FAQ 2: What is the difference between assent and consent?
Consent is a legally valid, fully informed, and voluntary agreement provided by an adult or emancipated minor [29]. Assent is the child's affirmative agreement to participate, demonstrating a willingness to engage beyond mere compliance with directions [31]. It involves explaining the study in age-appropriate language and seeking the child's agreement, even if their understanding of the risks and benefits is incomplete [29] [9].
FAQ 3: How should I handle a situation where a child dissents but their parent has consented?
A child's dissent—verbal or non-verbal—should be taken seriously and honored [29]. Exceptions are rare and ethically complex, typically considered only when the research offers a net direct benefit to the child that is not otherwise available and the intervention is necessary for their health [8]. Document all instances of dissent and the research team's response.
FAQ 4: Is financial compensation for participation considered coercive?
Compensation for time and effort is appropriate, but it must not reach a level that unduly influences a family's decision to enroll or remain in a study [8]. The compensation should be proportional to the burden of participation and should not be presented as a primary benefit.
FAQ 5: What should I do when a minor participant turns 18 during the study?
When a participant turns 18, you must obtain legally effective informed consent from them as an adult for any ongoing interactions or interventions [30]. The previously obtained parental permission and child assent are not sufficient. The IRB can provide guidance on this transition, including potential waivers for continued analysis of existing data [30].
Developmental Considerations for Assent Procedures
Table 1: Age-Appropriate Assent Strategies and Behavioral Markers
| Age Group | Developmental Capacity | Recommended Assent Strategies | Potential Dissent Cues |
|---|---|---|---|
| Infants & Toddlers (0-3 years) | Limited verbal understanding; communicates through behavior, emotion, and body language [31]. | Defer to parent/guardian on child's typical behavior; gauge comfort; allow for flexible scheduling (breaks, feeding, play) [31]. | Crying, fussing, turning away, pushing objects away, tantrums [31]. |
| Preschool (3-6 years) | Emerging language; can understand simple explanations; limited concept of future [31]. | Use simple, concrete language; short explanations; visual aids (e.g., smile/frown faces); offer limited choices [31] [32]. | Physical resistance, hesitation, silence, seeking proximity to caregiver [29]. |
| School-Age (7-11 years) | Growing reasoning ability; understands cause/effect; can articulate simple preferences [31]. | Use written assent forms at a low reading level (e.g., 2nd-3rd grade); explain procedures using familiar examples; check for understanding [31]. | Verbal "no", shaking head, asking to leave, discomfort, distraction [29]. |
| Adolescents (12-17 years) | Capacity for abstract thought; understands risks/benefits; longer attention span [31]. | Provide a detailed written assent form (~6th-grade level) similar to adult consent; emphasize voluntary participation and confidentiality [31] [30]. | Verbal refusal, questioning the purpose, declining to sign the form [29]. |
Table 2: Core Ethical Principles and Their Application to Pediatric Assent
| Ethical Principle | Definition | Practical Application in Assent Processes |
|---|---|---|
| Respect for Persons | Acknowledging the autonomy and agency of individuals [8]. | Treating the child as an active participant; honoring their assent or dissent; using age-appropriate communication [29]. |
| Beneficence | The obligation to maximize benefits and minimize harms [8]. | Designing studies with minimal risk; using child-friendly settings and procedures; ensuring the research is scientifically necessary [9]. |
| Justice | Fair distribution of the benefits and burdens of research [8]. | Ensuring equitable selection of participants; not exploiting vulnerable children; providing fair access to potential research benefits [9]. |
Experimental Protocols for Implementing Assent
Protocol 1: Establishing a Dynamic Assent Process
A one-time signature is insufficient. Ethical assent is an ongoing, relational process that requires continuous attention [32].
- Pre-Visit Preparation: Share child-friendly study information with the parent and child beforehand, if possible [33].
- Initial Agreement: Conduct the formal assent discussion at the beginning of the visit using IRB-approved materials and language [33].
- Continuous Check-ins: Before starting new tasks or procedures, briefly re-explain what will happen and check for willingness. Use phrases like, "Is it still okay if we do this next?" [31].
- Attunement to Non-Verbal Cues: Continuously observe for signs of distress or withdrawal (e.g., silence, fidgeting, avoidance). View these as potential dissent and be prepared to pause or stop [29] [32].
- Documentation: Document the initial assent and any instances of ongoing dissent or withdrawal in the research record [33].
Protocol 2: Integrating Play-Based and Multimodal Methods
For young children, traditional question-and-answer assent is often inadequate. Instead, use methods that align with how children naturally communicate [32].
- Create a Playful Environment: Set up a research space that allows for movement and interaction with familiar or interesting materials [32].
- Use Conceptual Play: Allow children to explore research tools (e.g., mock MRI scanners, puzzles) in a play-based way before data collection begins. This helps them understand and negotiate their participation [32].
- Observe Multimodal Cues: Recognize that assent and dissent are communicated through gesture, gaze, movement, silence, and interaction with peers or objects, not just words [32].
- Allow for Child-Led Pacing: Let the child's engagement and comfort level shape the pace and flow of the research session, even if it deviates from a rigid protocol [32].
The following diagram illustrates this continuous, multi-modal assent process.
Table 3: Research Reagent Solutions for Pediatric Assent
| Tool or Resource | Function/Purpose | Example Application |
|---|---|---|
| Age-Stratified Assent Forms | Written documents explaining the study in language tailored to specific developmental levels (e.g., ages 7-11, 12-17). | For school-age children, use short sentences and pictures; for adolescents, use a more detailed form at a 6th-grade reading level [31] [30]. |
| Visual Aids & Scales | Images, diagrams, or emoji scales (e.g., smiley to frowny faces) to help children understand procedures and express feelings. | To help a preschooler indicate their comfort level during a procedure or to explain the steps of a blood draw [31] [32]. |
| Microsampling Techniques | Medical equipment and methods that minimize the volume of blood drawn from a child. | Using a pediatric micro-sample tube instead of a standard vial to reduce discomfort and risk of iatrogenic anemia [9]. |
| Child-Life Specialists | Professionals trained in child development who help children cope with medical and research procedures. | A specialist uses a doll to demonstrate a procedure, teaches coping techniques, or provides distraction during a venipuncture [9]. |
| Digital Tools (VR, eBooks) | Technology to create engaging, low-threat environments for explaining studies and collecting data. | Using a virtual reality story to simulate the research experience before asking for assent [32]. |
| Modeling & Simulation Software | Computer software used to optimize trial design and reduce the number of participants needed. | Applying population PK modeling to obtain maximal information with fewer blood samples and participants [9]. |
Employing Child-Friendly Language and Visual Aids
Frequently Asked Questions
Q1: How can I check if the colors in my diagram are accessible for children with low vision?
A1: Ensure all text elements meet minimum color contrast ratios. Use automated tools like the axe accessibility engine to test contrast. For standard text, aim for a contrast ratio of at least 4.5:1; for large text (18pt or 14pt bold), a ratio of 3:1 is acceptable [34] [35].
Q2: What is the simplest way to create a text-based alternative for a complex flowchart? A2: Use nested lists with "If X, then go to Y" language to represent different paths or decisions, similar to a "choose your own adventure" book. This structure is inherently linear and easier for screen readers to navigate [36].
Q3: My diagramming tool only exports individual shapes. How can I make the entire chart accessible? A3: Combine the entire flowchart into a single high-quality image and provide a comprehensive text alternative. Describe the overall relationship and structure of the chart in the alt text, as you would explain it over the phone [36].
Q4: How do I properly set text color in a Graphviz node to ensure readability?
A4: Explicitly set both the fillcolor for the node's background and the fontcolor for the text. Do not rely on default colors, and always test the contrast ratio between your chosen colors [37].
Troubleshooting Guides
Problem: Low Color Contrast in Visual Materials
Issue: Text in diagrams, charts, or informational graphics does not have sufficient contrast against the background, making it difficult for children with low vision or color vision deficiencies to read.
Solution:
- Test Color Contrast: Use a color contrast analyzer to check the ratio between foreground (text) and background colors. Free browser extensions like the
axeDevTools can automate this on web content [34]. - Apply Minimum Ratios:
- Choose High-Contrast Colors: Select color pairs from the approved palette that naturally have high contrast, such as dark gray on white (#202124 on #FFFFFF) or white on a dark, vibrant color.
- Manually Verify in Graphics Software: When creating static images, use the contrast checker built into many design applications or consult contrast ratio tables.
Problem: Complex Flowchart is Difficult to Describe
Issue: A multi-layered, branching flowchart is challenging to convey in a text description, and the visual tool creates multiple individual images instead of one.
Solution:
- Simplify the Diagram: Consider breaking down one complex flowchart into several simpler, more focused diagrams [36].
- Create a Single Image: Export the entire flowchart as one high-resolution image. This can be done using the tool's export function or by taking a high-quality screenshot [36].
- Write Comprehensive Alt Text: Craft alternative text that describes the chart's purpose and overall structure. For example: "A flowchart describing the steps a child would take in our study. The full text description is provided in the section below the image." [36]
- Provide a Full Text Alternative: Publish a text version using headings or ordered lists below the visual chart. This benefits all users, not just those using assistive technology [36].
Color Contrast Requirements for Visual Aids
The table below summarizes the minimum contrast ratios required by the Web Content Accessibility Guidelines (WCAG) for text in visual materials.
| Text Type | Definition | Minimum Contrast Ratio | Enhanced Contrast (Level AAA) [38] |
|---|---|---|---|
| Standard Text | Text smaller than 18pt (24px) or not bolded 14pt (19px) | 4.5:1 [35] | 7:1 [38] |
| Large Text | Text that is 18pt (24px) or larger, or bolded 14pt (19px) or larger | 3:1 [35] | 4.5:1 [38] |
Experimental Protocol: Creating an Accessible Visual Aid
This protocol details a method for creating and validating a child-friendly visual aid that is both understandable and accessible.
Objective: To translate a complex research concept into a visual diagram that is accessible to children, including those with visual impairments.
Materials:
- Diagramming software (e.g., Graphviz, Mermaid)
- Color contrast analyzer (e.g., axe DevTools browser extension)
- Approved color palette
Methodology:
- Content Storyboarding:
- Define the key message and break it down into 3-5 core steps or ideas.
- Write a plain-language narrative for each step using short words and active voice.
- Visual Drafting:
- Create a basic flowchart or diagram layout, focusing on a clear logical flow.
- Node Styling: For each node, explicitly define both
fillcolorandfontcolorfrom the approved palette to ensure high contrast [37]. - Connection Lines: Use solid, dark-colored arrows (#5F6368 or #202124) on light backgrounds (#F1F3F4 or #FFFFFF).
- Accessibility Validation:
- Run the color contrast analyzer on the final diagram to check all text elements.
- If the diagram is digital, check that it is a single, tagged image and not a group of separate shapes.
- Text Alternative Creation:
- Develop a text-based version using a nested list structure that mirrors the diagram's logic [36].
- Place this text version directly below the visual aid in the document or webpage.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Child-Centric Research |
|---|---|
| Color Contrast Analyzer | Software tool to verify that text and background colors meet accessibility standards, ensuring materials are legible for children with low vision [34]. |
| Text-Based Outline | A structured text document using headings and lists to plan the logical flow of a visual aid before it is drawn [36]. |
| Diagramming Software (DOT Language) | A tool like Graphviz that uses a script-based language to generate consistent, reproducible diagrams where colors and styles can be precisely controlled [37]. |
| Approved Color Palette | A predefined set of accessible colors that ensures visual consistency and compliance with contrast ratios across all research materials. |
Accessible Diagram Specification
Diagram: Study Participation Steps
Diagram: Simple Consent Decision
Training Research Staff in Pediatric Communication and Ethics
Frequently Asked Questions (FAQs) and Troubleshooting Guides
This technical support resource addresses common challenges and procedural questions related to ethical communication and consent processes in pediatric research.
FAQ Category: Informed Consent & Assent Processes
1. What are the most common errors in the pediatric informed consent process and how can we avoid them?
| Common Error Type | Specific Examples | Troubleshooting & Prevention Strategies |
|---|---|---|
| Content & Readability | Risks downplayed; compensation ambiguities; complex protocol titles; length of commitment unclear [39]. | Use readability formulas (e.g., Fry Graph) [10]; employ visual aids and animated videos [10]; pilot-test forms with parents. |
| Process & Signature Irregularities | Missing signatures; consent by unapproved staff; dates missing or inconsistent [39]. | Implement pre-enrollment checklists; maintain a signature and delegation log; verify all personnel are IRB-approved for consent tasks. |
| Document Management | Use of expired ICFs; lost or misfiled forms; failure to re-consent when required [39]. | Establish a system for tracking ICF version control and expiration dates; use secure, organized storage; create a protocol for identifying events that trigger re-consenting. |
2. How should we determine a child's capacity to assent, and what does a valid assent process entail?
A child's capacity for assent is not determined by age alone but by their age, maturity, and psychological state [8]. The process should be tailored to the child's developing abilities.
- Best Practice Workflow: The following diagram outlines a dynamic approach to obtaining and maintaining assent.
- Key Protocol Details:
- Information Exchange: The purpose is to inform the child about the study in a language they understand, enhancing decision-making between parents and the child [8] [39].
- Documenting Dissent: A child's refusal (dissent) should be respected, and overriding it is only justifiable in exceptional circumstances, such as when the research intervention offers a direct benefit crucial to the child's health that is not available otherwise [11].
- Evolving Process: Consent is negotiable and should be revisited as the study progresses and the child's understanding evolves [40].
3. How can we effectively communicate complex trial concepts (like randomization) to children and parents?
Investigations show that central research concepts are not uniformly understood by parents or older pediatric patients [39].
- Troubleshooting Strategy: Move beyond dense text. Utilize interactive computer technologies, staged informed consent (providing information over multiple sessions), and visual methods to convey complex ideas [41] [39].
- Verification Method: Instead of asking "Do you understand?", use open-ended, non-directive questions such as, "Can you explain to me in your own words what this study is about?" or "What does it mean to be 'randomized' in this study?" [39]. This verifies comprehension more effectively.
FAQ Category: Ethical Challenges & Risk Management
4. How do we assess and justify risk levels in pediatric studies, especially when there is no direct benefit?
Federal regulations provide a structured framework for risk assessment, categorizing research into four approval levels [8] [4].
| Regulatory Category (45 CFR 46) | Risk/Benefit Profile | IRB Approval Criteria & Justification Requirements |
|---|---|---|
| §46.404 | Research not involving greater than minimal risk. | Approval requires that risks are minimized and are proportionate to the knowledge gained. "Minimal risk" is defined as the probability of harm or discomfort being no greater than that encountered in daily life or during routine medical/psychological exams [8]. |
| §46.405 | Research involving greater than minimal risk but presenting the prospect of direct benefit to the individual child. | The risk is justified by the anticipated direct benefit to the child; the risk-benefit ratio is at least as favorable as that of alternative approaches [8] [4]. |
| §46.406 | Research involving greater than minimal risk and no prospect of direct benefit, but likely to yield generalizable knowledge about the child's disorder or condition. | The risk represents a minor increase over minimal risk; the research experience is reasonably commensurate with the child's actual medical, psychological, or social situation; the knowledge is of vital importance for understanding or addressing the child's condition [8] [4]. |
| §46.407 | Research that does not fit the above categories but provides an opportunity to understand, prevent, or alleviate a serious problem affecting children's health or welfare. | The research must be reviewed by a national panel of experts (e.g., the Secretary of HHS) and found to present a reasonable opportunity to further the public health [4]. |
5. What strategies can mitigate the risk of coercion when recruiting pediatric participants?
- Clarify Roles and Care: Researchers should explicitly state that refusing to enroll or withdrawing from the study will not affect the child's clinical care or the family's relationship with their healthcare team [8]. Some suggest that staff not directly involved in the child's clinical care make the initial approach to eliminate this concern [8].
- Manage Compensation: While families should be compensated for their time and effort, the compensation must not be so substantial as to become an undue influence, forcing a family to enroll or stay in a study against their better judgment [8].
- Ensure Voluntariness: The consent and assent processes must be presented in a manner that emphasizes the voluntary nature of participation, allowing sufficient time for consideration and ensuring that the decision is free from pressure [40].
FAQ Category: Building Researcher Competence
6. What core ethical principles must research staff be trained on for pediatric studies?
Training must be grounded in the three core principles of the Belmont Report [8] [4] and applied through a pediatric lens.
- Foundational Ethical Framework: The following diagram illustrates how core ethical principles translate into practical applications for pediatric research.
7. What are the key components of a researcher's "toolkit" for ethical pediatric communication?
| Toolkit Component | Function & Purpose | Practical Examples & Notes |
|---|---|---|
| Age-Appropriate Information Sheets | To convey study information in a manner that matches the child's cognitive and emotional developmental stage [42]. | Separate forms for parents/guardians, adolescents, school-age children, and young children using visuals and simple language. |
| Visual Aids & Electronic Tools | To enhance understanding of complex procedures and concepts, and to reduce anxiety [10] [41]. | Animated videos illustrating the protocol; diagrams of study visits; interactive apps that explain randomization. |
| Open-Ended Question Scripts | To verify the child's and parents' understanding of the study beyond a simple "yes" or "no" [39]. | "Tell me in your own words what we are asking you to do." "What are the possible good and bad things that could happen if you are in this study?" |
| Dissent Acknowledgment Protocol | To formally recognize and document a child's unwillingness to participate, ensuring their voice is heard and respected [11] [40]. | A defined process for recording verbal or behavioral dissent. A plan for how staff should respond (e.g., pausing procedures, consulting with parents). |
Ensuring Ongoing Consent and Re-assent Throughout the Study
Frequently Asked Questions (FAQs)
Q1: What is the difference between re-consent and ongoing assent?
- Re-consent is a formal process required by ethical guidelines and federal regulations. It involves obtaining a signed, updated consent form from participants (or their guardians) when significant changes occur in the study, such as new risks, modified procedures, or extended duration [43] [44].
- Ongoing Assent is an informal, continuous process of checking in with a child participant to ensure they understand and are still willing to participate. It involves age-appropriate dialogues and reaffirming their agreement at various stages, especially in long-term studies [44] [9]. It is a key part of maintaining an ethical relationship with the young participant [45].
Q2: Under what specific circumstances is formal re-consent required in a pediatric study? Formal re-consent is required in the following circumstances [43] [44]:
| Circumstance | Description | Key Considerations |
|---|---|---|
| Substantive Protocol Changes | Major modifications to study objectives, procedures, or confidentiality. | Impacts the participant's willingness to continue; must be clearly explained. |
| New Risk Information | Emergence of new data on potential risks or a change in the magnitude of known risks. | Must be communicated in a timely manner. |
| Change in Participant Status | A child participant reaches the age of majority (e.g., turns 18) during the study [43]. | Requires obtaining consent as an adult using the full, approved consent form. |
| Extended Study Duration | The study timeline is extended beyond what was originally agreed upon. | Participants should agree to the new time commitment. |
| New Procedures | Addition of new interventions or data collection methods. | Participants must be informed of and agree to the new procedures. |
Q3: How should we approach a child who initially assented but later seems distressed or unwilling to continue? A child's ongoing willingness, and particularly their dissent (objection or refusal), must be taken seriously. Except in rare cases where the research intervention is the child's best or only medical option, a child's refusal to participate must be respected [46] [11]. The ethical approach is to pause the research procedures, explore the reasons for the child's distress in an age-appropriate manner, and honor their decision to withdraw if they persist in their refusal [11].
Q4: What methods can be used to document the re-consent process? The method should match the significance of the new information [44]:
- Consent Addendum: A short document outlining only the new or revised information. Ideal for single, significant changes (e.g., a new potential risk).
- Full Revised Consent Form: A completely new consent form. Best when there are numerous changes to the study.
- Information Letter with Verbal Confirmation: A letter sent to participants describing the changes, followed by a phone call or discussion to confirm their continued participation. This is often sufficient for minor changes.
- Verbal Re-consent: For non-sensitive information, a verbal discussion documented in the study records may be approved by the IRB.
Q5: A key study team member is leaving. Do we need to re-consent all participants? Not always. Re-consent is typically required for significant changes to the research team that could impact participant data confidentiality or how study samples are handled [43]. A change in a primary investigator or a key clinician may be significant enough to require re-consent, while the departure of a junior data analyst may not. The Institutional Review Board (IRB) can provide the final determination.
Troubleshooting Guides
Issue: A Child Participant is About to Reach the Age of Majority
Problem: A minor enrolled in your long-term study will soon turn 18, the age of legal adulthood in their state. The original consent was provided by their parent/guardian, with the child providing assent.
Solution: This is a standard ethical requirement. You must plan for and execute a formal re-consent process [43] [44].
- Plan in Protocol: The study protocol should have a built-in procedure for identifying and re-consenting participants as they approach the age of majority.
- Obtain IRB Approval: Submit the full, adult version of the Informed Consent Form (ICF) for IRB review and approval.
- Schedule the Session: Schedule a consent session with the soon-to-be adult participant, without their parents, to emphasize their new autonomous role.
- Conduct the Re-consent: Walk the participant through the full ICF, ensuring they understand they are now providing their own legally valid consent.
- Document: The participant must sign and date the new ICF. Their signature now replaces the parental permission.
Issue: New Safety Information Emerges from an Interim Data Analysis
Problem: Your safety monitoring board identifies a new, low-frequency adverse event that must be communicated to current participants.
Solution: This new information could impact a participant's willingness to continue, so a re-consent process is required [43] [44].
- Assess Urgency & Scope: Determine how quickly the information needs to be communicated and whether it applies to all participants or only a specific subset (e.g., those actively receiving the drug).
- Choose a Method: For clear, concise communication of a single new risk, a Consent Addendum is often the most effective tool as it highlights the new information without the clutter of the entire consent form [44].
- Execute the Plan:
- Contact: Inform participants of the new finding.
- Explain: Provide a clear explanation, using the addendum as a guide, and allow time for questions.
- Document: Have the participant (or guardian) sign the IRB-approved addendum to document their acknowledgment and continued agreement.
The following workflow diagram visualizes the decision-making process for ongoing consent and re-assent as described in the troubleshooting guides and FAQs.
Issue: A Young Child is Unable to Verbally Assent Due to Communication Needs
Problem: Your participatory research includes children with complex communication needs (CCN) who cannot provide assent through standard verbal or written means.
Solution: Move beyond a one-time procedural event and embrace a relational, ongoing process of seeking assent [45].
- Use Alternative Methods: Employ non-verbal methods to communicate the study and gauge willingness, such as pictures, symbols, eye gaze, body language, and gestures [45].
- Interpret Engagement: View the child's ambivalent or iterative (dis)engagement throughout the research process as continuous communication of their assent or dissent.
- Monitor Continuously: The researcher must be attuned to the child's reactions during every interaction. A change in engagement (e.g., turning away, showing signs of distress) should be interpreted as potential dissent and addressed immediately.
- Involve a Specialist: Work with a speech and language therapist or a child life specialist to develop appropriate communication strategies.
The Scientist's Toolkit: Essential Materials for Ethical Consent Processes
This table details key resources for implementing and managing ongoing consent and re-assent.
| Item / Resource | Function in the Consent/Assent Process |
|---|---|
| IRB-Approved Consent Form Templates | Provides the legally and ethically approved foundation for all consent discussions and documentation. Must be used for initial consent and any full re-consent [44]. |
| Consent Addendum Template | A specialized document for clearly communicating and documenting specific changes (e.g., new risks) without re-presenting the entire consent form, minimizing participant confusion [44]. |
| Age-Appropriate Assent Scripts & Visual Aids | Tools (e.g., simplified scripts, diagrams, videos) to explain the study to children in a way they can understand, facilitating genuine assent rather than mere compliance [9] [47]. |
| Participant Tracking System (with age alerts) | A database or system with alerts to flag when a pediatric participant is approaching the age of majority, ensuring timely planning for re-consent [43] [44]. |
| Documentation Log | A standardized system (e.g., within the study's source documents) for recording all consent-related interactions, including verbal reaffirmations, phone calls, and formal re-consent events [44]. |
Overcoming Real-World Hurdles: Strategies for Complex Consent Scenarios
Mitigating Coercion and Undue Influence in Recruitment
Troubleshooting Guides
Guide 1: Managing Interpersonal Relationships in Recruitment
Problem: A researcher wishes to recruit their own students for a study, creating a potential power differential where students may feel pressured to participate.
Solution:
- Indirect Recruitment: Use flyers or general announcements rather than direct, personal communication to avoid putting pressure on specific individuals [48].
- Independent Consent Process: Have a researcher who does not have an interpersonal relationship with the students conduct the informed consent process. This ensures students do not feel their decision will impact their relationship with their teacher [48].
- Blinding and Data Anonymization: Use coded information collected by someone other than the involved researcher so participants are not identifiable to them, protecting participant confidentiality and voluntariness [48].
Guide 2: Determining Appropriate Participant Compensation
Problem: An Institutional Review Board (IRB) struggles to decide if the financial compensation offered in a study protocol constitutes an undue inducement.
Solution:
- Avoid "Gut Feelings": IRBs often wrestle with these terms and may rely on intuition. Instead, refer to established ethical frameworks to guide decisions [49].
- Apply a Wage-Payment Model: Consider a standardized, hourly wage for participants' time and burden, rather than a market-based model that ties payment to the level of risk. This model is often favored over reimbursement or market models to avoid undue influence [49].
- Justify the Amount: In the protocol, clearly explain how the compensation amount was determined and why it is appropriate and not excessive for the participant population and study procedures [49].
Guide 3: Obtaining Valid Consent and Assent in International Pediatric Research
Problem: A multi-national pediatric psychology study faces different legal and cultural expectations for consent, making a single protocol challenging.
Solution:
- Early Ethical Planning: Engage international collaborators from the project's inception to identify key differences in ethical requirements across countries [50].
- Identify Local Leaders: Consult with pediatric ethics thought and policy leaders in each country to understand local standards and procedures [50].
- Adapt Communication: Provide information and assent documents in age-appropriate language. For example, in Australia, young people may be asked to summarize the information sheet to ensure comprehension [50]. The table below summarizes key international differences in consent and assent ages.
Table 1: International Variations in Consent and Assent Practices in Pediatric Research
| Country | Consent Age (Adult) | Assent Considerations | Key Regulatory Notes |
|---|---|---|---|
| Australia | 16 years old [50] | 14-16 years is a "gray area"; age-appropriate language required [50] | Data must be kept until 7 years after publication or until the child turns 25 [50] |
| Canada | No strict legal age [50] | Decided by ability to understand, often 12-14 years old [50] | Tri-Council Policy Statement (TCPS) certification required [50] |
| Denmark | 15 years and above [50] | Parents should be informed if participant is a minor [50] | GDPR must be followed for data protection [50] |
| Hong Kong | 18 years old [50] | Information not provided in search results | Data must be anonymized and stored securely [50] |
Frequently Asked Questions (FAQs)
Q1: What is the difference between coercion and undue influence? A1: Coercion involves an overt or implicit threat of harm to obtain compliance. Undue influence, by contrast, often occurs through an offer of an excessive or inappropriate reward to obtain compliance. IRBs are required to ensure research minimizes both [49].
Q2: How should I handle the recruitment of employees in my workplace? A2: Special safeguards are needed. Research should not imply that participation gives an employee a competitive advantage or that refusal will incur a penalty. Documentation of permission from an authorized workplace signatory is often required, and activities should ideally occur outside the participant's workplace or regular work hours to maintain confidentiality [48].
Q3: What are the key ethical principles when enrolling colleagues, friends, or family members? A3: You must consider how the research relationship could affect your personal relationship, especially if a study-related harm occurs, sensitive information is shared, or the participant withdraws. You must also ensure that providing compensation to these individuals complies with your institution's Conflict of Interest policies [48].
Q4: What are the unique considerations for obtaining consent in pediatric populations? A4: Children cannot legally provide informed consent. Therefore, you must obtain permission from a parent or guardian. Additionally, you must seek the child's assent—their affirmative agreement to participate—if they are mature enough to understand the study (typically around age 7 and older). The child's refusal to participate should generally be respected [51].
Q5: My study involves pediatric patients in a clinical setting. How can I mitigate therapeutic misconception? A5: When researchers are also clinicians for the patient, it creates a risk that the patient or parent may confuse research with therapy. To mitigate this, justify in the protocol why the clinician-researcher must be involved in recruitment and consent. If not justified, a clinician or staff member not affiliated with the research should conduct these processes [48].
Methodologies & Workflows
Experimental Protocol: Implementing a Coercion-Mitigated Recruitment Workflow
This protocol outlines a systematic approach to recruiting participants where a power differential exists (e.g., teacher-student, employer-employee).
1. Pre-Recruitment Phase
- Protocol Documentation: In the research protocol, explicitly identify any potential interpersonal relationships and power differentials. Provide a scientific justification for including these populations and detail the mitigation strategies [48].
- IRB/EC Review: Submit the detailed protocol, including all consent and assent forms, for review and approval by an Institutional Review Board or Ethics Committee [51].
2. Recruitment Phase
- Indirect Recruitment Launch: Use indirect methods such as flyers, online postings, or announcements made by a neutral party to advertise the study without targeting individuals directly [48].
- Independent Screener: Have an individual not involved in the research relationship conduct the initial screening of potential participants [48].
3. Informed Consent Phase
- Independent Consent Administrator: A member of the research team who has no interpersonal relationship with the participant conducts the entire informed consent process [48].
- Assent for Pediatrics: For child participants, use age-appropriate language to explain the study and obtain their assent. Ensure they understand they can withdraw at any time [51].
4. Data Collection and Management Phase
- Anonymization: Immediately de-identify data by using codes. Research personnel with an interpersonal relationship should not have access to the key that links codes to identities [48].
- Secure Storage: Store data according to institutional and international standards (e.g., password-protected, encrypted drives). Adhere to required data retention periods, which for pediatric data may extend for many years [50].
The following diagram illustrates this workflow:
Research Reagent Solutions
Table 2: Essential Resources for Ethical Recruitment
| Item / Solution | Function in Ethical Research |
|---|---|
| Indirect Recruitment Materials | Flyers, online postings, and general announcements used to recruit without targeting individuals directly, minimizing perceived pressure to participate [48]. |
| Age-Appropriate Assent Scripts | Pre-prepared, simplified explanations of the research study tailored to different child developmental stages (e.g., 7-11, 12-17) to ensure comprehension [50] [51]. |
| Independent Consent Administrator | A designated member of the research team with no pre-existing relationship to the participant, responsible for conducting the informed consent process to protect voluntariness [48]. |
| Data De-identification Protocol | A standardized procedure for immediately replacing personally identifiable information (PII) with a unique code after consent, safeguarding participant confidentiality [48]. |
| Ethical Compensation Framework | A pre-approved, standardized model (e.g., wage-payment model) for determining participant payment that minimizes the risk of undue inducement [49]. |
Addressing Therapeutic Misconception in Parents and Children
Technical Support Center: Troubleshooting Guides and FAQs
Frequently Asked Questions (FAQs)
Q1: What is therapeutic misconception in the context of pediatric research? Therapeutic Misconception (TM) occurs when parents or participants conflate the goals of clinical research with those of individual clinical care. In pediatric research, this often manifests when parents, hoping for direct medical benefit for their child, fail to understand that the primary purpose of research is to generate generalizable knowledge, not to provide individualized therapy. This is particularly prevalent in areas like neuroimaging research and trials for severe childhood diseases, where parental hope and the complexity of the science can obscure this fundamental distinction [52] [53].
Q2: Why is therapeutic misconception a critical ethical problem? TM compromises the validity of informed consent. If parents do not understand that procedures (like additional scans or placebo arms) are for data collection and not for their child's direct benefit, their consent is not fully informed. This undermines the ethical principle of respect for persons and can exploit the vulnerability of families desperate for new treatments [52] [53].
Q3: What are the common signs that a parent may be experiencing therapeutic misconception? Key indicators include:
- A belief that their child's treatment regimen will be individually tailored for optimal benefit within the study.
- An expectation that the researchers, who are also physicians, are functioning primarily as their child's doctor.
- Difficulty understanding the purpose of research-specific procedures like randomization, blinding, or the use of placebos.
- Expressing certainty of direct benefit from an experimental intervention whose efficacy is, in fact, the very question the trial is designed to answer [52] [53].
Q4: How can researchers assess the level of therapeutic misconception during the consent process? Implement a structured Therapeutic Misconception Assessment (TMA) during consent discussions. This involves asking open-ended questions to evaluate understanding, such as:
- "How is being in this study different from the regular medical care your child receives?"
- "Can you explain to me what parts of the procedure are done purely for research?"
- "What do you believe is the main reason we are doing this study?" [52] Systematically documenting responses helps identify and address misconceptions.
Q5: What is the difference between consent and assent in pediatric research? Consent is the legally valid permission provided by a parent or legal guardian for their child to participate in research. Assent is the affirmative agreement to participate obtained from the child themselves, using age-appropriate language. Ethical research requires both, wherever possible. The American Academy of Pediatrics suggests that children as young as 7 years old should be given the opportunity to assent or decline participation [8] [9].
Troubleshooting Guide: Addressing Key Challenges
Problem 1: Parental Hope Leading to Misconception
- Scenario: A parent of a child with a rare, life-limiting condition consents to a Phase I trial, expressing unwavering belief that the experimental drug will cure their child, despite explanations about the trial's primary goal of safety and dosing.
- Solution:
- Reframe the Goal: Emphasize that the study is a "first step" to help future children, managing expectations from the outset.
- Transparent Communication: Clearly and repeatedly state the likelihood of direct benefit (e.g., "We hope this might help, but the main reason for this study is to see if the drug is safe and how the body handles it, so we can learn for the future.").
- Involve a Neutral Educator: Utilize a research subject advocate or a consent professional not directly involved in the child's clinical care to reduce perceived pressure [8] [53].
Problem 2: Coercion and Undue Influence
- Scenario: A family feels pressured to enroll because their primary pediatrician, whom they trust, is also the principal investigator. They fear that refusing may harm their relationship and the quality of their child's care.
- Solution:
- Explicit Reassurance: Proactively and explicitly state, both verbally and in the consent form, that the decision to participate or withdraw will not affect the regular clinical care or the patient-provider relationship.
- Separation of Roles: Where feasible, have a member of the research team who is not the treating clinician approach the family for consent [8].
- Review Compensation: Ensure that compensation for participation (for time and travel) is not so high as to be coercive, but is fair and does not cover medical procedures [8].
Problem 3: Inadequate Child Assent
- Scenario: A 9-year-old child remains silent when asked for assent but later expresses fear and reluctance about blood draws to a nurse.
- Solution:
- Developmentally Appropriate Dialogue: Use simple, concrete language. For a school-age child, explain what will happen, what they will feel, and how long it will take.
- Assess Understanding: Ask the child to explain the study back in their own words.
- Respect Refusal: Honor the child's dissent whenever possible, especially in non-beneficial research. Overriding a child's refusal should be a rare exception, considered only when there is a clear and direct net benefit to the child that outweighs their discomfort [8] [9].
Problem 4: Complexity of Study Design
- Scenario: Parents struggle to understand the concept of randomization and a double-blind, placebo-controlled trial design.
- Solution:
- Use Analogies: Explain randomization by comparing it to flipping a coin. Clarify that "blinding" means neither the family nor the doctors know which treatment is received to ensure the results are fair and not based on expectations.
- Simplify Documents: Craft the Informed Consent Form at a mandatory 8th-grade reading level, avoiding complex medical jargon [8].
- Visual Aids: Utilize diagrams and flowcharts to illustrate the study design and different patient pathways.
Quantitative Data on Pediatric Research and Consent
The table below summarizes key regulatory risk categories and prevalence data relevant to addressing therapeutic misconception.
Table 1: Regulatory Risk Categories and Ethical Considerations in Pediatric Research
| CFR Reference | Risk Category | IRB Approval Criteria | Relevance to Therapeutic Misconception |
|---|---|---|---|
| 45 CFR 46.404 [8] | Research not involving greater than minimal risk. | Approved if adequate provisions are made for soliciting assent and parental permission. | Low risk of TM, but clear communication about non-therapeutic procedures remains critical. |
| 45 CFR 46.405 [8] | Research involving greater than minimal risk but presents the prospect of direct benefit to the individual child. | Approved if the risk is justified by the anticipated benefit; the relation of risk to benefit is at least as favorable as alternatives. | High risk of TM. Parents may overestimate the prospect of benefit. Clarity on the uncertainty of the benefit is key. |
| 45 CFR 46.406 [4] | Research involving greater than minimal risk and no prospect of direct benefit, but likely to yield generalizable knowledge about the child's disorder/condition. | Approved if the risk represents a minor increase over minimal risk and the research experiences are commensurate with those inherent in the subject's situation. | Very high risk of TM. Must be unequivocally clear that the child will not benefit directly from the procedures. |
| Contextual Data | Prevalence & Impact | ||
| Drug Approval Gap [8] | ~80% of drugs prescribed to children have not been studied in this population. | This gap creates a backdrop of urgency and hope that can fuel TM. | |
| Incentive Effectiveness [8] | Legislative incentives (e.g., BPCA) alone have been insufficient to bridge the pediatric research gap. | Highlights the systemic pressure for pediatric research, within which ethical consent must be managed. |
Experimental Protocol: Assessing and Mitigating Therapeutic Misconception
Objective: To systematically identify and reduce the level of therapeutic misconception in parents during the informed consent process for a pediatric clinical trial.
Methodology:
- Pre-Consent Baseline Assessment:
- Before the formal consent discussion, provide parents with a one-page summary of the study's purpose.
- Administer a short, anonymized questionnaire to gauge initial understanding, using Likert-scale and open-ended questions (e.g., "What is the main goal of this study?").
Structured Consent Intervention:
- Utilize a consent script that includes "TM Checkpoints." At these points, the researcher pauses to ask and document understanding.
- Key Checkpoints:
- After explaining the study's purpose: "So, just to be clear, could you tell me in your own words what the main goal of this research is?"
- After explaining randomization: "How would you explain to a family member why your child's treatment is assigned by chance?"
- After explaining procedures: "Can you identify which tests/scans are part of standard care and which are done purely for research?"
- After discussing risks/benefits: "What do you see as the potential benefits for your child from participating? What are the potential drawbacks?"
Post-Consent Evaluation:
- After the consent discussion and a 24-hour reflection period, administer a follow-up validated TM assessment tool.
- The tool should be designed to distinguish between therapeutic misestimation (underestimating risk/overestimating benefit) and therapeutic misconception (fundamental misunderstanding of the research purpose) [53].
Data Analysis and Documentation:
- Analyze responses to identify persistent areas of misunderstanding.
- Document the process and findings for IRB review and for refining future consent processes.
Workflow Diagram: Therapeutic Misconception Mitigation Protocol
The diagram below outlines a systematic workflow for integrating therapeutic misconception assessment and mitigation into the pediatric research consent process.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools for Ethical Pediatric Research Consent
| Tool / Solution | Function in Addressing Ethical Challenges |
|---|---|
| Validated TM Assessment Scales | Quantitative and qualitative instruments to measure the prevalence and depth of therapeutic misconception in a study population. Provides empirical data for IRBs. |
| Age-Appropriate Assent Forms | Customized documents and visual aids designed to explain the research process to children at different developmental stages (e.g., < 7, 7-12, >12 years). |
| IRB-Approved Consent Scripts | Standardized scripts with built-in TM checkpoints to ensure all research staff deliver consistent, complete, and ethically robust information. |
| Decision Aids | Visual aids, such as flowcharts or booklets, that help parents understand complex concepts like randomization, placebo control, and the difference between research and clinical care. |
| Digital Recording Systems (with consent) | To record consent discussions (with permission) for quality assurance and for IRB audit purposes, ensuring adherence to the protocol. |
| Real-World Data (RWD) Analytics | Data from claims, lab tests, and electronic health records used to design more efficient and less burdensome trials, thereby minimizing risk and discomfort for children [54]. |
Adapting to Acute vs. Non-Acute Healthcare Settings
Frequently Asked Questions (FAQs)
Q1: What are the core ethical principles governing pediatric research in any healthcare setting? The conduct of pediatric research is guided by three core ethical principles established by the Belmont Report: Respect for Persons, Beneficence, and Justice [4]. Respect for Persons requires obtaining informed consent and assent appropriately. Beneficence obligates researchers to maximize benefits and minimize harms. Justice demands a fair distribution of the burdens and benefits of research [4].
Q2: How does the definition of "minimal risk" influence protocol design across settings? Minimal risk is a foundational concept, defined as the probability and magnitude of harm or discomfort being no greater than those ordinarily encountered in daily life or during routine medical/psychological examinations [8]. This definition directly influences what studies an Institutional Review Board (IRB) can approve, especially for research that does not offer a direct benefit to the child [8]. The assessment of risk must be carefully justified in your protocol.
Q3: What are the specific regulatory categories for approving pediatric research? An IRB can only approve pediatric research if it meets the conditions of one of the categories outlined in 45 CFR 46, Subpart D [4]. The following table summarizes these categories:
| 45 CFR 46 Category | Regulatory Criteria for Approval | Common Application Scenarios |
|---|---|---|
| §46.404 | Research not involving greater than minimal risk [4]. | Non-invasive behavioral studies, retrospective chart reviews. |
| §46.405 | Research involving greater than minimal risk but presenting the prospect of direct benefit to the individual child [8]. | Clinical trials of a new therapeutic drug or device. |
| §46.406 | Research involving greater than minimal risk and no prospect of direct benefit, but likely to yield generalizable knowledge about the child's disorder or condition [8]. | Pathophysiological studies involving slightly more than minimal risk biopsies or imaging. |
| §46.407 | Research that does not fit the above categories but presents an opportunity to understand, prevent, or alleviate a serious problem affecting children's health or welfare [8]. | Studies requiring national-level IRB or Department of Health and Human Services review. |
Q4: How should the informed consent process be adapted for adolescents? Adolescents with mature decision-making capacity should be provided with a comprehensive study overview and their assent should be obtained separately from their parent's permission [8]. The American Academy of Pediatrics suggests that a waiver of parental permission can be considered for minimal-risk research that addresses questions specific to the adolescent population, or when the research involves treatments for which minors can legally provide their own consent [8].
Q5: What specific strategies prevent coercion during participant recruitment? To prevent coercion, researchers must explicitly state that refusal to participate or subsequent withdrawal will not affect the child's clinical care or relationship with their healthcare provider [8]. Some institutions recommend that the initial approach for a study be made by staff not directly involved in the child's clinical care to eliminate perceived pressure [8]. Furthermore, while compensation for time and effort is appropriate, it must not be so high as to become a coercive influence [8].
Experimental Protocol: Consent Process Workflow
The following diagram maps the critical decision points and ethical safeguards in the pediatric consent and assent process, applicable across acute and non-acute settings.
Research Reagent Solutions: Essential Materials for Ethical Protocol Implementation
| Item/Tool | Function in Ethical Research Conduct |
|---|---|
| IRB-Approved Informed Consent Form (ICF) | The primary document ensuring regulatory compliance. It must contain all required elements (21 CFR 50), be written at an 8th-grade reading level, and be approved by the IRB before use [8]. |
| Age-Adapted Assent Forms | A suite of documents and scripts designed to explain the study to children of different developmental stages in language they can understand, facilitating their voluntary agreement [8]. |
| Institutional Review Board (IRB) | An independent ethics committee that reviews, approves, and monitors research involving human subjects to ensure ethical conduct and regulatory adherence [4]. |
| Documentation of Consent Process | A secure system (e.g., regulatory binder) for storing signed consent/assent forms and notes documenting the consent process, as required by ICH-GCP guidelines [8]. |
| Coercion Mitigation Script | A standardized script for researchers to use during recruitment that explicitly states that refusal to participate will not affect the child's medical care [8]. |
Within the sensitive realm of pediatric research, obtaining valid consent is a cornerstone of ethical practice. Traditional "opt-in" models, requiring active, explicit agreement, can sometimes lead to low participation rates and research samples that are not representative of the broader patient population [55]. This guide explores the implementation of "opt-out" consent procedures as a flexible alternative for specific, lower-risk research contexts. When designed and deployed correctly, opt-out methods can streamline participation while upholding the core ethical principles of respect for persons, beneficence, and justice, particularly crucial when research involves children and adolescents.
FAQs & Troubleshooting Guides
FAQ 1: What is the fundamental ethical difference between opt-in and opt-out consent?
- Answer: The difference lies in the default action and the requirement for affirmative agreement.
- Opt-In: Requires an explicit, affirmative action by the individual (or their legal representative) to agree to participate. Silence or inaction is interpreted as a "no." This model prioritizes explicit, prior authorization [56].
- Opt-Out: Individuals are informed that they will be included in the research unless they take a specific action to decline. Silence or inaction is interpreted as a "yes." This model assumes consent by default but provides a mechanism for refusal [57].
FAQ 2: Under what conditions might an opt-out approach be ethically acceptable in pediatric research?
- Answer: An opt-out approach may be considered when several conditions are met, often for research that is lower risk and of high public health value. Key conditions include:
- The research presents no more than minimal risk to participants [58].
- The use of an opt-in procedure is impracticable or would lead to a low or selective participation rate that threatens the study's validity [55].
- The research has the potential to generate significant public health benefits, such as improving care quality for a wide population of children [55].
- Robust safeguards are in place, including pseudonymization of data and oversight by a Research Ethics Committee (REC) [55] [57].
- Parents and, where appropriate, children are provided with clear, accessible information about the research and the opt-out process [59].
FAQ 3: A parent claims they never received information about the study yet their child's data was included. How should this be addressed?
- Troubleshooting Steps:
- Verify Communication Channels: Immediately audit the methods used to disseminate information (e.g., letters, patient portal messages, clinic posters) to confirm they were deployed correctly.
- Review Documentation: Check your records to confirm the information was sent to the parent's last known address or preferred contact method.
- Prioritize the Request: If the parent's concern is valid, apologize for the oversight and act immediately. Facilitate a simple and immediate opt-out process.
- Rectify the Situation: Assure the parent that their child's data will be removed from the study dataset and will not be used in future analyses.
- Improve Processes: Use this incident to review and strengthen your communication and tracking systems to prevent future occurrences.
FAQ 4: Our opt-out study is experiencing a higher-than-expected objection rate. What could be the cause?
- Troubleshooting Steps:
- Evaluate Clarity of Information: Review the information sheets and notices. Is the purpose of the research, the data usage, and the opt-out method explained in simple, understandable language? Complex or legalistic language can cause anxiety and lead to opt-outs [59].
- Assess Opt-Out Mechanism: Is the method for opting out clear, simple, and accessible? If the process is burdensome, it can cause frustration, but a poorly explained simple process can also be problematic.
- Gather Feedback: Consider surveying a small group of parents to understand their perceptions and concerns. There may be misunderstandings about the research's risks or benefits.
- Check External Factors: Look for negative media coverage or misinformation circulating in patient communities that could be eroding trust [57].
The following tables summarize key quantitative findings from research on consent procedures, which can inform the design and expectations for an opt-out model.
Table 1: Comparison of Consent Rates between Opt-In and Opt-Out Procedures
| Consent Procedure | Average Consent Rate | Key Findings |
|---|---|---|
| Opt-In | 84.0% (across multiple studies) [55] | Lower consent rates can limit the pool of participants and the generalizability of findings. |
| Opt-Out | 96.8% (in a single study) [55] | Significantly higher participation rates, leading to larger and potentially more representative datasets. |
| Direct Comparison (One Study) | Opt-In: 21.0%Opt-Out: 95.6% [55] | Highlights the dramatic difference in participation that can result from the choice of consent model. |
Table 2: Demographic Biases Associated with Opt-In Consent Procedures
| Demographic Factor | Bias in Opt-In Consenters |
|---|---|
| Sex/Gender | More likely to be male [55]. |
| Education Level | Higher levels of education [55]. |
| Income & Socioeconomic Status | Higher income and higher socioeconomic status [55]. |
Experimental Protocols
Protocol 1: Designing and Implementing an Ethical Opt-Out Workflow
This protocol outlines the key steps for setting up an opt-out consent process for a minimal-risk pediatric study using electronic health records (EHR).
1. Pre-Implementation Ethics Review * Submit a detailed application to your Institutional Review Board (IRB) or Research Ethics Committee (REC). The application must justify the use of an opt-out model based on minimal risk, impracticability of opt-in, or risk of consent bias [55] [58]. * Present the comprehensive communication plan and the simple opt-out mechanism for approval.
2. Develop Family-Centric Communication Materials * Create information sheets and notices using clear, jargon-free language appropriate for the general public. * Visually distinguish these research communications from standard clinical documents. * Explicitly state: the research purpose; what data will be used; that participation is presumed unless they opt-out; how to opt-out; and who to contact with questions [59].
3. Multi-Channel Communication Deployment * Direct Communication: Send letters or secure electronic messages via patient portals to the parents/guardians. * In-Clinic Information: Display clear notices in waiting areas and examination rooms. Provide leaflets at reception desks. * Staff Training: Ensure all frontline clinical staff are aware of the study and can direct parents to the correct information and opt-out process.
4. Implement a Simple and Accessible Opt-Out Mechanism * Provide multiple easy channels for opting out, such as a toll-free number, a dedicated email address, a reply form, or a prominent link in the patient portal. * The action required to opt-out should be a single, straightforward step.
5. Data Management and Compliance * Maintain a secure, real-time "do-not-contact" list that is integrated with the research dataset. * Establish a process to immediately flag and remove data for any individual who opts out, ensuring ongoing compliance. * Document all steps for audit purposes.
Protocol 2: Evaluating Representativeness and Consent Bias
This methodology allows researchers to assess whether the opt-out model successfully mitigated the biases typically associated with opt-in approaches.
1. Pre-Study Population Analysis * In collaboration with data custodians, generate an anonymous profile of the entire eligible pediatric population for key demographic variables (e.g., age, sex, socioeconomic status indices, geographic location, primary diagnosis).
2. Post-Enrollment Comparator Analysis * Once the final research cohort is established (after the opt-out window has closed), generate a similar anonymous demographic profile for the study participants.
3. Statistical Comparison for Bias * Compare the demographic profile of the research cohort against the profile of the total eligible population. * Use statistical tests (e.g., chi-square tests for categorical variables, t-tests for continuous variables) to identify any significant differences. * Success Metric: A well-functioning opt-out system should show no significant demographic differences between the participants and the total eligible population, indicating a representative sample has been achieved [55].
Visual Workflows
Opt-Out Consent Workflow for Pediatric Research
Mechanisms of Consent Bias in Research
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Components for Implementing an Electronic Opt-Out System
| Tool / Component | Function in Consent Management |
|---|---|
| Digital Asset Management (DAM) Platform | A centralized system to store consent forms and related digital assets, link them to specific participants, and automate reminders for consent expiry or need for re-consent (e.g., when a child comes of age) [60]. |
| eConsent Platform with API Integration | A specialized software solution (e.g., Standard Health Consent - SHC) that can be embedded into patient portals or health apps. It provides clear interfaces for information and manages consent metadata securely [57]. |
| Identity Management System (e.g., Keycloak) | An open-source system that handles user authentication, creates unique user pseudonyms, and can integrate with national health IDs, ensuring secure and private access to consent platforms [57]. |
| Electronic Signature Solution (e.g., DocuSign) | For studies requiring documented consent or authorization, these tools provide a secure and verifiable method for obtaining electronic signatures that meet legal standards [58]. |
| Tiered Opt-Out Module | A software component that moves beyond a simple binary choice, allowing users to express nuanced preferences (e.g., agreeing to public health research but objecting to commercial use). This enhances autonomy and trust [57]. |
Managing Cultural, Educational, and Language Barriers
Frequently Asked Questions (FAQs)
Q1: What are the most common system-level barriers to including participants with limited English proficiency (LEP) in research? Researchers consistently report that a lack of dedicated funding for interpretation and translation services is a primary system-level barrier [61]. This is often compounded by a low institutional prioritization of language access, leading to a lack of clear best practices and standardized protocols for obtaining informed consent across language differences [61].
Q2: How can I adapt the informed consent process for communities with low literacy or different cultural understandings of research? The consensus is to move beyond a simple written form. Best practices include [62] [63]:
- Using verbal or alternative consent procedures tailored for low-literacy settings.
- Simplifying terminologies and avoiding complex medical jargon.
- Involving community members in the development and translation of consent documents to ensure they are accurate, respectful, and contextually appropriate.
- Engaging in ongoing community consultations to build trust and align the research process with local cultural norms.
Q3: What are the key technical requirements for delivering telehealth services to participants in their homes? Establishing a reliable clinic-to-home service requires attention to three core computer systems, with the following minimum specifications [64]:
Table 1: Technical Specifications for Telehealth Services
| Computer System | Component | Minimum Specification | Rationale |
|---|---|---|---|
| Connectivity | Internet Speed | 1 Mbps download/upload | Necessary for adequate video quality, especially with movement. |
| Hardware | Computer | Laptop with i5 processor, 4 GB RAM, USB ports | Meets minimum requirements for videoconferencing software. |
| Hardware | Audio/Video | External webcam (e.g., Logitech c920) and headset | Ensures clear audio for coaching and protects confidentiality. |
| Software | Videoconferencing | Platform like Skype | Cost-effective and widely accessible for participants. |
Q4: How do language barriers specifically impact the quality of pediatric clinical encounters? Physicians report that language barriers fundamentally change the encounter, leading to [65]:
- Communication Challenges: A loss of nuanced communication, including tone and humor, which impedes relationship-building.
- Structural Inefficiencies: Encounters feel more rushed and chaotic, with less time for in-depth discussion or "hand-on-the-door" concerns.
- Assessment Limitations: It can be more difficult to assess patient development and obtain a detailed history, as the process is often diluted through an interpreter.
Troubleshooting Guides
Issue: Low Comprehension of Informed Consent Among Participants
Problem: Participants, particularly in multicultural or low-literacy settings, may not fully understand the research study's purpose, procedures, risks, and benefits, even after going through the consent process [63].
Solution: Implement a Multi-Layered Consent Verification Protocol
- Simplify the Language:
- Rewrite the consent form using simple, clear language at a lower reading level.
- Replace complex scientific jargon with locally understandable terms identified through community engagement [63].
- Utilize Verbal Explanation and Visual Aids:
- Use diagrams, flowcharts, or pictograms to illustrate the study design and participant journey.
- Train research staff to explain the study verbally using a standardized script.
- Employ a "Teach-Back" Method:
- After explaining a key concept, ask the participant to explain it back in their own words.
- This is not a test of the participant, but a check of the researcher's ability to explain clearly.
- Document the Process:
- Meticulously document the steps taken to ensure comprehension, including the use of interpreters and community validators, for ethics committee review [62].
Issue: Inconsistent Use of Interpreters by Research Staff
Problem: Even when interpreter services are available, research staff may underutilize them for certain tasks or lack the confidence to use them effectively, compromising the quality of communication [66] [65].
Solution: Establish a Standardized Interpreter Use Protocol with Training
- Develop a Mandatory Use Policy:
- Clearly define situations that require a professional interpreter (e.g., obtaining consent, discussing medical history, explaining complex procedures) [66].
- Provide Practical Training:
- Move beyond simply informing staff that interpreters are available.
- Implement hands-on training sessions that simulate triadic conversations (researcher-interpreter-participant) and cover best practices, such as speaking directly to the participant and using short, clear sentences [65].
- Integrate with Workflow:
- Ensure the process for booking an interpreter is simple and efficient, ideally integrated into the research team's scheduling software to minimize delays.
Experimental Protocols
Protocol 1: AI-Driven Translation and Synthesis for Multilingual Educational Content
This protocol details a methodology for creating linguistically and culturally tailored educational videos for healthcare providers and caregivers, as demonstrated in a pediatric care study [67].
- Objective: To translate and voice-narrate an original English video curriculum into target languages while preserving contextual meaning and clinical accuracy.
- Materials:
- Procedure:
- Transcription: Transcribe the original English narrative using a dictation tool (e.g., in Microsoft Word) [67].
- AI Translation: Use the generative language model (GPT-4) to translate the transcript into the target language [67].
- Expert Review: Have native-speaking medical professionals review the translated text for clinical accuracy, tone, and cultural relevance [67].
- Voice Synthesis:
- Video Integration: Render the synthesized audio as an mp3 file and integrate it into the original video using editing software, adjusting playback speed as needed for synchronization [67].
Protocol 2: Community-Engaged Development of Culturally Appropriate Consent Forms
This protocol outlines a qualitative approach to identifying and addressing language-related barriers in the informed consent process, based on research in Uganda [63].
- Objective: To identify community perspectives on informed consent terminology and create study documents that are clear, accurate, and respectful.
- Materials:
- Recruitment support from local community leaders.
- Guide for Focus Group Discussions (FGDs).
- Audio recording equipment.
- Qualitative data analysis software (e.g., NVivo).
- Procedure:
- Participant Selection: Purposively sample a diverse group of stakeholders, including community members, past research participants, and Community Advisory Board (CAB) members, representing various language groups [63].
- Focus Group Discussions: Conduct segregated FGDs using open-ended questions to explore experiences and challenges with consent forms, including comprehension of terminology and procedural understanding [63].
- Thematic Analysis: Transcribe and code the data to identify emergent themes, such as specific medical terms that are difficult to translate or social norms that affect consent [63].
- Community Validation: Hold workshops with a diverse group of stakeholders (researchers, participants, local leaders) to present findings and reach a final agreement on the preferred terminology and consent process [63].
- Tool Implementation: Revise the informed consent documents and procedures based on the validated findings.
Research Reagent Solutions
Table 2: Essential Resources for Overcoming Language and Cultural Barriers
| Item / Solution | Function / Application | Specific Examples / Notes |
|---|---|---|
| Professional Interpreter Services | Facilitates accurate, real-time communication between researcher and participant during consent and data collection. | Essential for ensuring genuine informed consent and protecting against medical error [66] [65]. |
| Generative Language Models (GLMs) | Aids in initial translation of study materials (e.g., consent forms, surveys) into multiple languages. | GPT-4 can be used for translation but requires review by native speakers for clinical and cultural accuracy [67]. |
| Voice Synthesis Software | Generates synthetic, human-like voiceovers for educational video content in multiple languages. | Tools like the ElevenLabs library can create voice profiles from native speaker samples [67]. |
| Community Advisory Boards (CABs) | Provides ongoing, structured community engagement to ensure cultural appropriateness and build trust. | Members can include religious, cultural, and local political leaders who offer insights into local dialects and norms [63]. |
| Videoconferencing Platforms | Enables remote research consultations, coaching, and data collection (telehealth) to overcome geographic barriers. | Platforms must meet security (HIPAA) requirements and have sufficient bandwidth for behavioral observation [64]. |
Workflow and Pathway Diagrams
AI-Driven Content Localization
Multi-Level Strategy for Inclusive Research
Lessons from the Field: Case Studies and International Perspectives
Technical Support Center: Troubleshooting Guides and FAQs
This section provides targeted support for researchers navigating ethical and logistical challenges within a pediatric research registry.
Frequently Asked Questions (FAQs)
| Question Category | Common Issue | Evidence-Based Solution |
|---|---|---|
| Participant Recruitment | Low enrollment yield through gatekeeper organizations [68] | Develop a participant contact registry for direct access to willing families, bypassing administrative layers [68]. |
| Informed Consent | Complexity of consent with a vulnerable pediatric population [9] | Implement a dual process: obtain informed consent from a parent or legal guardian and seek affirmative assent from the child using age-appropriate language [9]. |
| Logistical Barriers | Families cite heavy daily demands as a barrier to participation [68] | Design protocols with maximum flexibility. Offer multiple participation windows, remote options, and compensate for time and travel to reduce burden [68]. |
| Trust & Engagement | Participants feel uninformed or undervalued after a study ends [68] [69] | Establish a transparent communication plan. Provide appropriate information about the study's purpose and, where feasible, a summary of findings to participants [68]. |
| Ethical Study Closure | Managing the termination of a study, whether planned or abrupt [69] | Develop a participant-centered closure plan upfront. This includes communicating the conclusion, explaining the reason for termination, and honoring participants' contributions to uphold trust [69]. |
| Minimizing Risk | Anxiety or distress in child participants during procedures [9] | Use child-friendly settings and trained staff. Employ distraction techniques, allow caregiver presence, and use topical anesthetics for blood draws to minimize harm and discomfort [9]. |
Troubleshooting Common Experimental Protocol Challenges
| Protocol Stage | Challenge | Recommended Methodology |
|---|---|---|
| Assent & Consent | Ensuring ongoing, meaningful assent from a maturing child in a long-term study [9]. | Re-consenting Process: Integrate periodic re-assent conversations. For adolescents transitioning to adulthood, establish a clear protocol for obtaining their own informed consent [9]. |
| Data Collection | Balancing the need for robust data with the ethical imperative to minimize participant burden and risk [9]. | Micro-sampling Techniques: Utilize micro-analytical methods and population PK modeling to drastically reduce blood draw volumes and frequency in clinical trials [9]. |
| Upholding Ethics in Practice | Unexpected ethical dilemmas arise during the research process that were not anticipated in the procedural ethics approval [70]. | Reflexive Care-Ethical Approach: Foster continuous ethical mindfulness. Engage in collective reflexivity with the research team to navigate "ethically important moments" as they occur [70]. |
Research Reagent Solutions: Essential Materials for Ethical Pediatric Research
The following table details key non-physical "reagents" or frameworks essential for conducting ethical research within a pediatric registry.
Table: Essential Frameworks and Tools for Pediatric Registry Research
| Item | Function in the Experimental Process |
|---|---|
| Participant Contact Registry | A pre-established database of families interested in research, streamlining recruitment and providing direct access, thus reducing gatekeeper burdens [68]. |
| Age-Stratified Protocol | A study design that ensures appropriate representation and analysis across different pediatric age groups (neonates, infants, children, adolescents) to account for developmental differences [9]. |
| Age-Appropriate Assent Scripts | Standardized, developmentally-tailored language and visuals used to explain the research study to a child, ensuring their agreement is meaningful and informed [9]. |
| Data Safety Monitoring Board (DSMB) | An independent committee of experts that provides ongoing oversight and review of clinical trial data to ensure participant safety and study integrity [9]. |
| Ethical Symmetry Principle | A conceptual framework that posits children should be accorded the same level of ethical respect as adults, though the practical application may differ. It mandates their active involvement in research to the extent of their abilities [70]. |
| Belmont Report Principles | The foundational ethical principles of Respect for Persons, Beneficence, and Justice. These guide the entire research process, from design to recruitment, ensuring voluntary participation, a favorable risk/benefit ratio, and fair selection of subjects [69] [70]. |
Experimental Protocol: The Anti-Coercion Workflow for Obtaining Consent and Assent
This detailed methodology outlines the steps for ensuring voluntary, informed, and ongoing participation in the TARGet Kids! Registry.
Key Steps in the Anti-Coercion Protocol:
- Initial Contact and Information Dissemination: Provide a clear, comprehensive information sheet about the registry's purpose, what participation involves, potential benefits and burdens, and data usage policies [68] [9].
- Dual-Layer Consent and Assent Process:
- Parental/Guardian Informed Consent: The parent or legal guardian provides formal, written consent after having all their questions answered satisfactorily. This is a legal and ethical requirement [9].
- Child Affirmative Assent: The child's willingness to participate is secured through an age-appropriate conversation. This is not a legal contract but an ethical imperative that respects the child as a person. For older children and adolescents, this process should be increasingly detailed [9].
- Ensuring Voluntariness and Right to Withdraw: Explicitly state, both at the outset and throughout the study, that participation is entirely voluntary. Participants and their families can withdraw at any time without any penalty or loss of benefits to which they are otherwise entitled [9]. This is a critical safeguard against coercion.
- Ongoing Process and Re-assent: For long-term registries, consent and assent are not one-time events. Researchers must periodically check in with families and, importantly, with the growing child. As a child matures, their understanding deepens, and the assent process should be revisited. For adolescents approaching the age of majority, a re-consenting process should be implemented to obtain their own informed consent [9].
- Transparency and Communication: Maintain open channels of communication. Inform participants about the registry's findings and how their contribution is advancing science. This fosters trust and reinforces the collaborative nature of the research partnership [68] [9].
Obtaining valid informed consent in pediatric research is a cornerstone of ethical practice, yet it presents distinct challenges in low-resource settings. In Malawi, where limitations in healthcare access and research literacy prevail, understanding the nuanced experiences of guardians and research staff is critical for improving ethical standards. This technical support guide synthesizes qualitative insights from hospital-based pediatric research studies in Malawi, providing researchers, scientists, and drug development professionals with evidence-based troubleshooting guidance. The content is framed within a broader thesis on ethical challenges in pediatric research consent, aiming to bridge the gap between international ethical standards and local realities. The following sections provide a structured analysis of common challenges, practical solutions, and methodological protocols to strengthen the consent process in similar contexts.
Troubleshooting Guide: FAQs on Pediatric Consent Challenges in Malawi
FAQ 1: What are the primary contextual barriers to obtaining meaningful informed consent in Malawi? Research in Malawi identifies several interconnected barriers. Guardians often associate research participation with therapeutic benefits (therapeutic misconception), a perception observed across both acute and non-acute study settings [71]. Parental stress due to a child's illness heightens vulnerability, potentially compromising understanding and decision-making capacity [71]. Furthermore, socio-cultural factors, such as decision-making norms that require spousal consultation, can complicate consent when only one parent is present at the hospital [71]. Low research literacy and challenges in distinguishing research from clinical care further exacerbate these issues [71].
FAQ 2: How does the research setting (acute vs. non-acute) impact the consent process? The research setting significantly influences guardian comprehension and recall. Studies show that guardians in non-acute settings were more likely to recall information shared during the consent process compared to those in acute settings [71]. The high-stress environment of acute care, often involving emergency conditions, can overwhelm guardians, limiting their capacity to absorb complex research information [71]. This necessitates tailored consent approaches that account for the specific pressures of the clinical context.
FAQ 3: What challenges do research staff face during consent procedures? Research staff reported feeling pressured to recruit participants, which sometimes led to an emphasis on study benefits over risks [71]. In acute settings, staff encountered problems when guardians were alone and unable to consult their absent partners, a step often required for collective decision-making [71]. Navigating the fine line between thorough information disclosure and the practicalities of recruitment in a low-resource, high-burden environment remains a key challenge for frontline staff.
FAQ 4: What practical solutions can enhance understanding and ethical rigor? Stakeholders in Malawi have proposed several facilitator strategies. The use of visual materials can aid in explaining complex research concepts [72]. Enhanced community engagement and the use of patient advocates help build trust and demystify research goals within the community [72]. Furthermore, principal investigators are advised to define minimum process requirements and conduct study-specific training for staff to ensure consistency and quality in consent procedures, moving away from a one-size-fits-all approach [71].
Data Synthesis: Comparative Analysis of Consent Experiences
The table below summarizes key quantitative and qualitative findings from the research, highlighting the contrasting experiences across different settings and roles.
Table 1: Summary of Qualitative Insights from Pediatric Research in Malawi
| Aspect | Findings from Acute Settings | Findings from Non-Acute Settings |
|---|---|---|
| Guardian Comprehension | Lower recall of study information due to stress and urgent medical situations [71]. | Higher likelihood of recalling information shared during the consent process [71]. |
| Decision-Making Process | Guardians felt the role of absent spouses was neglected; staff faced challenges consenting solo guardians [71]. | Not reported as a prominent issue. |
| Therapeutic Misconception | Prevalent among guardians, who associated research participation with direct medical benefit [71]. | Prevalent among guardians, who associated research participation with direct medical benefit [71]. |
| Staff Approach | Staff reported pressure to recruit, sometimes emphasizing benefits over risks [71]. | Staff reported pressure to recruit, sometimes emphasizing benefits over risks [71]. |
Table 2: Demographic Profile of Hospital Guardians at Kamuzu Central Hospital, Malawi
| Demographic Characteristic | Finding | Implication for Consent Process |
|---|---|---|
| Gender | Predominantly female (83%) [73]. | Consent materials and communication should be accessible to women. |
| Median Age | 37.8 years [73]. | Information should be tailored to adults of varying ages. |
| Literacy | High illiteracy rate (72%), determined by inability to sign one's name [73]. | Heavy reliance on verbal explanation and visual aids; written forms are insufficient. |
| Primary Occupation | Self-employed (95%), often as rural farmers (63%) [73]. | Concepts should be explained using familiar, non-technical analogies. |
Experimental Protocols: Methodologies for Qualitative Inquiry
Protocol 1: In-Depth Interviewing for Eliciting Guardian and Staff Experiences
- Objective: To explore the nuanced views, experiences, and challenges faced by guardians and research staff in the informed consent process for pediatric research.
- Study Setting: Conducted in the pediatric department and research wards of a large referral hospital in Southern Malawi (e.g., Queen Elizabeth Central Hospital), encompassing both acute (inpatient) and non-acute (outpatient) settings [71].
- Participant Recruitment:
- Data Collection:
- Conduct 20 or more in-depth interviews using a semi-structured guide with open-ended questions [71].
- Interviews should be audio-recorded, conducted in the local language (e.g., Chichewa), and later transcribed verbatim and translated into English for analysis [71].
- Ensure interviews occur in a private setting to encourage candid responses [71].
- Data Analysis:
- Utilize thematic analysis to identify, analyze, and report patterns (themes) within the data [71].
- Use software such as NVivo 11 to facilitate coding and organization of data [71].
- Compare findings across different study types (e.g., clinical trial vs. observational) and settings (acute vs. non-acute) to identify contextual differences [71].
Protocol 2: Systematic Assessment of Consent Comprehension
- Objective: To evaluate participants' understanding of key study information after the consent process is complete.
- Method: Following the standard consent discussion, employ a short, structured questionnaire or a set of open-ended questions to assess comprehension of critical elements [71].
- Key Assessment Areas:
- Purpose of the research (distinguishing it from routine care).
- Voluntary nature of participation and the right to withdraw.
- Key procedures and their risks.
- Potential benefits (clarifying if there is no direct therapeutic benefit).
- Randomization and placebo use, if applicable [74].
- Troubleshooting: If misunderstanding is identified, the consent process should be revisited and clarified immediately. This may require multiple, shorter consent sessions.
Visualizing the Workflow: Consent Process Challenges and Solutions
The diagram below maps the logical flow of challenges encountered in the pediatric consent process in Malawi and the corresponding evidence-based solutions.
The Scientist's Toolkit: Essential Reagents for Qualitative Research
For researchers aiming to conduct similar qualitative studies on consent processes, the following "reagents" or methodological tools are essential.
Table 3: Research Reagent Solutions for Qualitative Consent Studies
| Research 'Reagent' | Function & Application | Example from Malawian Studies |
|---|---|---|
| Semi-Structured Interview Guides | To ensure consistent exploration of key topics while allowing flexibility to probe emergent themes. | Guides with open-ended questions for guardians and staff about their consent experiences [71]. |
| Bilingual Data Collection Tools | To ensure linguistic and cultural appropriateness, enabling participation in local languages. | Questionnaires translated into Chichewa and back-translated for quality assurance [73]. |
| Digital Audio Recorders | To capture interviews verbatim for accurate transcription and thematic analysis. | Used in in-depth interviews to record guardian and staff responses [71]. |
| Qualitative Data Analysis Software (e.g., NVivo) | To facilitate efficient coding, organization, and analysis of large volumes of textual data. | NVivo 11 software was used to manage and thematically analyze interview transcripts [71]. |
| Comprehension Assessment Checklists | To systematically evaluate participants' understanding of core consent concepts post-disclosure. | A tool to check understanding of voluntariness, risks, and procedures [71]. |
Comparative Analysis of Consent and Assent Policies Across Countries
The international character of pediatric psychology and clinical research has grown significantly, bringing a strong imperative for researchers to understand and navigate the differing regulations that govern consent and assent across national boundaries [50]. Conducting ethical research with children, a vulnerable population, requires a complex interaction among researchers, child participants, and their legal guardians to ensure the research is appropriate, safe, and legally compliant [50]. This complexity is magnified in international projects where legal requirements and societal expectations for assent, consent, and permission vary widely between participating nations [50]. Finding a way to anticipate and address such differences in approval requirements and processes at early stages of project development can significantly smoothen and expedite project approval, ultimately advancing global child health research while maintaining the highest ethical standards.
Global Regulatory Frameworks and Definitions
Foundational Ethical Principles
The ethical basis for protecting child participants in research stems from several foundational documents and principles. The Belmont Report, issued in 1978, served as the basis for core regulations applicable to all federally supported, conducted, or regulated research involving humans in the United States [75]. Internationally, the Declaration of Helsinki provides guidance that for a legally incompetent minor, the investigator must obtain informed consent from the child's legally authorized representative in accordance with applicable law [42]. Even a legally incompetent minor may be able to give assent to decisions about participation in research, and in such cases the investigator must obtain that assent in addition to the consent of a legally authorized representative [42].
General principles of medical ethics applied to pediatric research include [42]:
- Respect for life, human dignity, and personal autonomy
- Beneficence (do some good)
- Non-maleficence (do no harm)
- Justice
Key Terminology and Definitions
Table: Essential Consent and Assent Terminology
| Term | Definition | Application Context |
|---|---|---|
| Informed Consent | Voluntary agreement given by a person with legal capacity to participate in research after understanding the facts, implications, and future consequences | Typically provided by parents or legal guardians for minors |
| Assent | A child's affirmative agreement to participate in research, beyond mere failure to object | Obtained from children with developing capacity, alongside parental consent |
| Permission | Agreement by parents or guardians for a child to participate in research | Legal authorization for participation |
| Capacity | Ability to understand the research purpose, procedures, risks, and benefits, and appreciate the implications of participation | Assessed individually, not solely by age |
| Legally Authorized Representative | Individual authorized under applicable law to consent on behalf of a prospective participant | Varies by jurisdiction, typically parents or legal guardians |
Comparative Analysis of National Policies
Age Requirements and Capacity Assessment Across Countries
Table: International Comparison of Consent and Assent Requirements
| Country | Legal Age of Consent | Assent Considerations | Special Requirements |
|---|---|---|---|
| Canada | No defined legal age; determined by capacity to understand | Capacity assessed individually; children as young as 12-14 may consent if capable | Tri-Council Policy Statement (TCPS 2) certification required; criminal record checks often needed |
| Australia | 16 years (14-16 is a gray area) | Parent- and child-appropriate information sheets required; young people asked to summarize information | "Working With Children Check" required in some states; data kept until 7 years after publication or child turns 25 |
| Denmark | 15 years and above | Parents should be informed if participants are minors | GDPR compliance required for data storage; schools and clinics require director approval |
| Hong Kong | 18 years | Information provided to minors alongside parental consent | Anonymized data analysis; password-protected computers; locked rooms for data storage |
| United States | Varies by state; federal regulations categorize research risk levels | Requirement varies based on child's maturity and psychology | Subpart D of 45 CFR 46 provides additional protections; IRB must include pediatric expertise |
The United States federal regulations governing research with children establish a framework based on four categories of research [75]:
- Research not involving greater than minimal risk
- Research involving greater than minimal risk but presenting the prospect of direct benefit to individual subjects
- Research involving greater than minimal risk and no prospect of direct benefit to individual subjects, but likely to yield generalizable knowledge about the subject's disorder or condition
- Research not otherwise approvable that presents an opportunity to understand, prevent, or alleviate a serious problem affecting the health or welfare of children
Documentation and Process Requirements
In Canada, where there is no defined age requirement to consent children into research, ethical guidelines indicate that children of any age can consent to research studies if they have the capacity to understand and appreciate the significance of the research and the implications of the risks and benefits to themselves [76]. Capacity is typically assessed by asking a child to explain the purpose, procedures, risks, and benefits of the research in their own words and acknowledge their voluntary choice to participate in research [76].
The European Paediatric Psychology Network and Confederation of European Specialists in Paediatrics (CESP) recommend that separate age-appropriate information sheets and consent or assent forms should be developed for the parents or legal representatives and for the child [42]. A child should be able to withdraw willingly from any research project without detriment [42].
Methodological Approaches and Experimental Protocols
Innovative Consent Methodologies: Video Consent Protocol
A recent study conducted at The Hospital for Sick Children in Toronto compared video consent with traditional written informed consent in pediatric rheumatology research [76]. The experimental protocol was designed as follows:
Research Question: Does video consent improve participant understanding, satisfaction, and preference compared to written informed consent in pediatric research?
Study Design: Randomized cross-over trial where participants were randomized to receive either video consent or written informed consent first for a registry study.
Participants: 99 caregivers and 76 patients randomized into video consent (n = 88) and written informed consent (n = 87) groups.
Intervention Details:
- Video Consent: Pre-recorded audio supported by an animated/live action video explaining the research study, followed by questions with a research team member.
- Written Informed Consent: Traditional paper consent form with verbal discussion with research team member.
Assessment Measures:
- Comprehension questionnaire (Max score = 12)
- Satisfaction questionnaire (Max score = 5)
- Time to complete consent process
- Preference between methods
Results:
- Comprehension was high in both groups (medianvideo consent = 11 and medianwritten informed consent = 10) with no significant difference
- Satisfaction was high in both groups (medianvideo consent = 4 and medianwritten informed consent = 5) with no significant difference
- Video consent took slightly longer (median 408 seconds vs 360 seconds)
- Decisive evidence for participant preference for video consent (BF10 = 2.307x10^11)
Video Consent Methodology Workflow
Digital Recruitment and Engagement Protocols
A 2023 cross-sectional survey study in Chongqing, China investigated public perspectives on digitalizing pediatric research, revealing important considerations for modern research protocols [77]. The methodology included:
Survey Development: Questionnaire refined based on literature and clinical experience, then revised and culturally adapted by 7 pediatric doctors with diverse professional backgrounds.
Participant Recruitment: 4,231 valid questionnaires from caregivers of kindergarten children (aged 2-7 years), with 25.1% from urban areas and 74.9% from suburban areas.
Digital Implementation: Web-based survey using "Lediaocha" platform with 50 mandatory questions across 4 pages, accessible through WeChat on smartphones.
Key Findings:
- 38.3% of caregivers recognized the growing popularity of digital tools
- 36.7% supported using digital tools in pediatric research
- Only 25.2% favored online-only research methods
- Main concerns: privacy issues (77.4%) and potential addiction (58.1%)
- Preferred recruitment: public accounts of research institutions (80.4%)
- Preferred contact: telephones (62.3%) and social media apps (58.6%)
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Essential Resources for Pediatric Consent Research
| Tool/Resource | Function/Purpose | Application Context |
|---|---|---|
| Age-Appropriate Information Sheets | Explain research procedures in language suitable for child's developmental level | Required for assent process; separate versions for parents and children |
| Capacity Assessment Protocol | Standardized method to evaluate child's understanding of research | Used when no fixed age of consent; involves explaining purpose, risks, benefits |
| Video Consent Materials | Pre-recorded audio-visual explanations of research studies | Alternative to written consent; improves accessibility for varying literacy |
| Digital Survey Platforms | Web-based tools for collecting participant perspectives and data | Enables broader recruitment; platforms must ensure data privacy |
| Multi-Language Consent Documents | Translated materials for diverse populations | Essential for international research and multicultural populations |
| IRB/REB Submission Templates | Standardized forms for ethics committee approval | Streamlines ethical review process across institutions |
| Data Security Systems | Password protection, encryption, secure storage | Protects participant confidentiality; required by GDPR and other regulations |
Troubleshooting Guide: FAQs for Common Research Challenges
Recruitment and Retention Challenges
Q: What strategies can improve recruitment and retention in pediatric research, especially in underrepresented populations? A: Research indicates that suburban caregivers may have more positive impressions of pediatric research and face fewer participation barriers compared to urban caregivers [77]. Effective strategies include implementing a hybrid online-offline follow-up approach, using both official institutional channels and unofficial social media for recruitment, and directly addressing privacy concerns that represent the primary barrier for 77.4% of caregivers [77]. Offering flexible participation methods and clear communication about data protection can significantly improve recruitment and retention.
Q: How can researchers address the primary concerns of caregivers regarding pediatric research participation? A: The main concerns include privacy issues (77.4%), potential addiction to digital tools (58.1%), and fears about children being "experimental subjects" (70.9-76.6%) [77]. Researchers should implement transparent data handling procedures, define clear time limits for digital interaction, and emphasize the educational and health benefits of participation. Providing opportunities for children to ask questions and withdraw willingly can alleviate concerns about coercion.
Consent Process Implementation
Q: What methodology should be used when assessing a child's capacity to consent in jurisdictions without defined age requirements? A: In countries like Canada where capacity determines consent ability rather than a specific age, researchers should develop a standardized assessment protocol [76]. This typically involves asking the child to explain in their own words: the research purpose, procedures involved, potential risks and benefits, and their understanding that participation is voluntary [76]. Documentation of this assessment is crucial for ethical and regulatory compliance.
Q: How can researchers improve comprehension in the consent process while maintaining efficiency? A: The video consent study demonstrated that while video consent took slightly longer (48 seconds more than written consent), it was strongly preferred by participants without sacrificing comprehension [76]. Researchers should consider implementing multimedia approaches for complex concepts while providing concise written summaries for reference. The increased time investment may be justified by improved participant experience and preference.
International Collaboration Challenges
Q: What strategies can facilitate ethical approval for international pediatric research collaborations? A: International surveys identify several key strategies: begin ethical planning from the project's inception; identify pediatric ethics thought and policy leaders in each country; confirm pertinent policies and procedures in each location; and develop a common terminology for ethics discussions across borders [50]. Establishing these foundations early can significantly smoothen and expedite the multi-country ethics approval process.
Q: How should researchers handle varying age requirements for consent and assent across countries? A: When operating in multiple jurisdictions, researchers must comply with the most restrictive requirements across all participating countries. The research protocol should document the specific consent and assent procedures for each country, including appropriate age thresholds and assessment methods for capacity where applicable [50] [42]. Using a standardized matrix that compares requirements across all participating countries can help ensure compliance.
International Ethics Approval Workflow
The comparative analysis of consent and assent policies across countries reveals both significant variations and common ethical foundations in pediatric research. While national regulations differ in their specific age requirements and procedural details, there is universal recognition of children's vulnerability and the need for special protections [50] [75]. Emerging methodologies such as video consent and digital recruitment platforms offer promising approaches to enhance participant understanding and engagement while addressing practical challenges in research implementation [76] [77]. As international collaboration in pediatric research continues to grow, developing flexible yet rigorous approaches to consent and assent that can adapt to varying national requirements while maintaining the highest ethical standards will be essential for advancing child health research globally.
The Role of Ethics Committees and IRBs in Validating Protocols
Institutional Review Boards (IRBs): Gatekeepers of Research Ethics
Institutional Review Boards (IRBs), also known as Independent Ethics Committees (IECs) or Research Ethics Committees, are formally designated groups responsible for reviewing and monitoring biomedical research involving human subjects [78] [79]. Their fundamental purpose is to safeguard the rights, safety, and welfare of people who participate in clinical trials [78] [79]. This oversight function is particularly critical in pediatric research, where participants are considered a vulnerable population requiring additional protections [9] [80].
IRBs use a group process to review research protocols and related materials—such as informed consent documents and investigator brochures—ensuring that studies comply with ethical standards and regulatory requirements before they begin and through periodic continuing review [78] [79]. These committees have the authority to approve, require modifications in (to secure approval), or disapprove research [78].
Essential IRB Functions and Operations
IRBs must follow detailed written procedures for four key operations as mandated by HHS and FDA regulations [81]:
- Conducting initial and continuing review of research.
- Determining which projects require review more often than annually.
- Ensuring prompt reporting to the IRB of proposed changes in research activity and ensuring that changes are approved by the IRB before implementation.
- Ensuring prompt reporting of any unanticipated problems involving risks to subjects or others, any instance of serious or continuing noncompliance, and any suspension or termination of IRB approval [81].
The following workflow details the IRB protocol validation process:
IRB Review Criteria for Pediatric Research Protocols
When validating protocols, especially for pediatric studies, IRBs evaluate research against a specific set of ethical and regulatory criteria. The foundational principles of Respect for Persons, Beneficence, and Justice, as outlined in the Belmont Report, form the bedrock of this assessment [79] [80]. For research involving children, these principles are operationalized through additional safeguards.
The table below summarizes the risk categories defined in U.S. regulations (45 CFR 46§D) that guide IRB approval of pediatric research:
| Risk Category | Regulatory Definition | IRB Approval Requirements |
|---|---|---|
| Research not involving greater than minimal risk | The probability and magnitude of harm or discomfort are not greater than those ordinarily encountered in daily life or during routine physical/psychological examinations [8]. | Approval permitted if adequate provisions are made for soliciting assent from children and permission from parents/guardians [8]. |
| Research involving greater than minimal risk but presenting the prospect of direct benefit to the individual subject | The risk is justified by the anticipated benefit to the subjects; the relation of the risk to benefit is at least as favorable as that presented by available alternative approaches [8]. | Approval permitted [8]. |
| Research involving a minor increase over minimal risk without the prospect of direct benefit | The risk represents a minor increase over minimal risk; the research is likely to yield generalizable knowledge about the subject's disorder or condition [8]. | Approval permitted only if the research presents experiences reasonably commensurate with those inherent in the subject's actual or expected medical, psychological, or social situations [8]. |
International Variations in Pediatric Consent and Assent
A significant challenge in international pediatric research is the variation in regulations governing consent and assent. A survey of pediatric psychology researchers across 14 countries highlighted key differences in practices and requirements [50] [82].
| Country / Region | Age of Consent/Assent Considerations | Key Local Requirements |
|---|---|---|
| Australia | 16 years old; 14-16 is a gray area [50]. | Data must be kept until 7 years after publication or until the child turns 25 (whichever is later) [50]. "Working With Children Check" required for researchers in some states [50]. |
| Canada | No fixed legal age for assent; decided by the ability to provide informed consent (as young as 12-14) [50]. | Tri-Council Policy Statement (TCPS) certification required; institutional standards for data storage (e.g., at least 5 years post-publication) [50]. |
| Denmark | 15 years old and above; parents should be informed if participants are minors [50]. | GDPR must be followed for data protection; data usually kept for 5-10 years [50]. |
| Hong Kong | 18 years old [50]. | Anonymized data analysis; data stored in a locked room on password-protected computers [50]. |
| United States | Children cannot give legally binding consent until age 18; parental permission is required. Assent is generally sought from children aged 7 and above [8] [80]. | Research must comply with FDA regulations (21 CFR 50, 56) and HHS regulations (45 CFR 46). IRB review is mandatory [78] [80]. |
| European Union | A legal representative (parent) must provide informed consent. Assent is sought from children considered mature enough to understand [80]. | Pediatric Investigation Plan (PIP) required for new medicines. Approved by a national ethics committee and the European Medicines Agency [80]. |
Troubleshooting Guide: Common IRB Challenges in Pediatric Research
FAQ: Informed Consent and Assent Process
Q: What are the essential elements of a valid assent process for a child? A valid assent process involves more than just a form; it is a continuous, interactive process. Researchers must explain the study in age-appropriate language that the child can understand, focusing on what the child will experience, potential discomforts, and the voluntary nature of participation. The child's affirmative agreement to participate should be sought, and their refusal (dissent) must be respected, unless the research holds out the prospect of direct benefit that is not available outside of the research context [8] [9] [80].
Q: How should researchers handle a situation where a parent consents but the child assents and then later dissents during the study? The ethical obligation to respect the child's ongoing willingness to participate is critical. If a child who is capable of assent later dissents—for example, by refusing a specific procedure or withdrawing from the study altogether—that decision should be honored, barring exceptional circumstances where the intervention is directly life-saving. Researchers should establish clear protocols for identifying and responding to dissent throughout the study period [9].
Q: In international studies, how do we reconcile different national standards for consent and assent? The best practice is to designate an ethics lead within each country involved in the project. This person is responsible for confirming local policies and engaging with local ethics boards. The multinational team should then create a summary of ethical requirements by country and ensure that the research protocol complies with the most rigorous rules and regulations among all participating locations [82].
FAQ: Risk Assessment and Protocol Design
Q: What strategies can be used to minimize risks and discomforts for children in clinical trials? Protocols should be specifically designed to reduce physical and psychological distress. This includes:
- Physical Discomfort: Using topical anesthetics before needle sticks, utilizing indwelling catheters to reduce repeated venipunctures, employing microsampling techniques to minimize blood draw volumes, and establishing strict limits on total blood draw volume based on the child's weight and total blood volume [9] [80].
- Psychological Distress: Creating child-friendly settings, employing trained child-life specialists, allowing caregivers to be present during procedures, using distraction techniques, and explaining procedures in a non-threatening, age-appropriate manner [9].
Q: What is the role of a Data and Safety Monitoring Board (DSMB) in pediatric trials? A DSMB (or Data Monitoring Committee, DMC) is an independent group of experts that reviews accumulated interim data from a clinical trial to monitor participant safety and treatment efficacy. While the IRB focuses on the initial and periodic review of the overall study design and consent process, the DSMB has primary responsibility for detailed review of interim data and can recommend that a trial be stopped early due to safety concerns, clear efficacy, or futility [83] [9].
The Scientist's Toolkit: Essential Reagents for Ethical Pediatric Research
Beyond the scientific reagents, conducting ethically sound pediatric research requires a set of procedural and methodological "tools" to ensure compliance and participant protection.
| Tool / Solution | Primary Function | Application in Pediatric Research |
|---|---|---|
| Ethics Committee (IRB/IEC) Approval | Provides independent ethical oversight and formal approval to conduct the research. | Mandatory gateway before study initiation; ensures the protocol adheres to ethical principles and regulatory standards [78] [79]. |
| Informed Consent/Permission Forms | Documents the process of providing key information to parents/legal guardians, enabling them to give permission for the child's participation. | Must be written at an appropriate reading level (e.g., 8th grade); must include all required elements of 21 CFR 50.25/ICH-GCP [8] [80]. |
| Age-Appropriate Assent Forms & Scripts | Tools to facilitate the child's understanding and affirmative agreement to participate. | Critical for respecting the child's autonomy; must be tailored to the child's developmental level (e.g., simplified text, pictures) [9] [82]. |
| Data Safety Monitoring Plan (DSMP) | A formal plan for monitoring the safety of participants and the validity and integrity of the data. | Outlines processes for adverse event reporting, interim data reviews, and may specify the use of an independent DSMB for higher-risk studies [83] [9]. |
| Cultural & Linguistic Adaptation Framework | A systematic approach to adapting research materials for different cultural and linguistic contexts. | Essential for international research; ensures consent/assent materials and protocols are respectful and comprehensible across different populations [50] [82]. |
| Microsampling/Low-Volume Assay Technologies | Technologies that enable diagnostic testing or pharmacokinetic analysis with very small blood volumes (e.g., dried blood spots). | Directly minimizes the risk of iatrogenic anemia and reduces discomfort for pediatric participants, aligning with the principle of minimizing harm [9] [80]. |
The following diagram illustrates the logical relationship between core ethical principles and their practical application in protocol validation:
Best Practices for Multi-National Pediatric Research Collaborations
FAQ: Building and Sustaining Effective Collaborations
Q: What are the key elements for establishing a successful multi-national pediatric research collaborative?
A successful collaborative requires a formalized structure and a commitment to shared learning. Key elements include:
- A Formal Charter: Establish a collaborative charter to define the mission, governance, and operational principles [84].
- Diverse Leadership: Incorporate leadership roles for both clinical professionals and family representatives to embed the patient and family voice at the highest level [84].
- Structured and Unstructured Learning: Combine formal activities like monthly meetings and annual summits with informal online forums and "subcommunities" for role-specific or topic-based discussion. This fosters both deep learning and trusted relationships [84].
- Multi-Institutional Research: Leverage the collaborative's scale to conduct robust, IRB-approved studies across multiple sites, generating generalizable new knowledge [84].
Q: How can our collaborative ensure it is truly embedding the patient and family perspective?
Beyond token representation, meaningful engagement requires dedicated resources and infrastructure.
- Compensate Family Partners: Financially compensate family advisors and patient representatives for their time and expertise [84].
- Create Exclusive Spaces: Establish subcommunities reserved solely for family advisors to learn from and empower one another privately [84].
- Scholarship Funds: Provide scholarships to support patient and family attendance at in-person summits, ensuring financial barriers do not limit participation [84].
- Co-Design: Involve patients and families in the development of research projects and manuscripts, offering them an opportunity to review and contribute insights [84].
Q: What is the biggest challenge in managing multi-site collaborations, and how can it be addressed?
A common challenge is the variation in data definitions and visualization tools across sites, which can lead to confusion and hinder accurate benchmarking [85].
- Adopt Standard Definitions: Use indicator definitions that adhere to international standards and recommended data sources [85].
- Cocreate Tools: Develop data visualization dashboards and profiles collaboratively across organizations to ensure consistency and mutual understanding [85] [86].
- Define Purpose and Audience: Clearly define the purpose, target audience, and added value of any new data visualization tool at the outset of a project [85].
Troubleshooting Common Collaboration Challenges
This section addresses specific operational issues you might encounter.
Problem: Inconsistent data across sites prevents meaningful analysis.
- Solution: Implement a structured data processing plan. Map data elements to common standards (like openEHR archetypes) and use standardized classification systems (e.g., International Classification of Childhood Cancer, Third Edition) to ensure all sites are processing and reporting data uniformly [86].
Problem: Lack of engagement from community partners or specific sites.
- Solution: Focus on building and maintaining trust. Allocate adequate staffing and funding for partnership management. Build on existing partnerships rather than always starting new ones, and create avenues for bidirectional communication to ensure all partners feel heard and valued [87].
Problem: High staff turnover disrupts the continuity of collaborative projects.
Essential Research Reagents & Solutions for Collaborative Pediatric Research
The following table outlines key non-laboratory "reagents" – the frameworks and tools essential for conducting ethical and effective multi-national pediatric research.
| Research Reagent / Solution | Function / Explanation |
|---|---|
| Collaborative Charter | A formal document that establishes the collaborative's mission, governance structure, and operational rules, providing a foundational framework for all activities [84]. |
| Patient & Family Advisory Council (PFAC) | A structured group of patients and family members that provides ongoing advice and guidance to ensure the research priorities and processes align with the community's needs [84]. |
| Multi-Site IRB Approval Protocol | A pre-established framework for navigating and harmonizing ethical reviews across different institutions and countries, streamlining the study launch process. |
| Harmonized Core Outcome Set | A standardized set of outcomes, developed with input from patients and providers across different cancer types, that ensures all studies are measuring and reporting the same clinically relevant endpoints [86]. |
| Quality Improvement Registry | A population registry of children receiving care for a specific condition, which provides a large, representative sample for high-quality data collection, feedback, and research [88]. |
| Data Visualization Dashboard | An interactive platform (e.g., using openEHR) that allows researchers and clinicians to visualize complex patient data, track outcomes, and facilitate benchmarking across sites [89] [86]. |
Experimental Protocol: Implementing a Multi-National Quality Improvement Study
This protocol outlines a methodology for conducting a collaborative quality improvement project, based on the model used by successful pediatric networks [84] [88].
Aim: To improve a specific clinical process (e.g., communication between doctors and nurses) across multiple pediatric hospitals.
Methodology:
- Topic Selection & Team Formation: Focus on a high-impact condition or safety issue. Form a multi-disciplinary improvement committee at each participating site, including clinicians, nurses, data analysts, and family representatives.
- Define Aims & Metrics: Set a clear, measurable overall aim. Use the Model for Improvement to define specific, measurable, achievable, relevant, and time-bound (SMART) goals. Establish a core set of metrics to track progress.
- Develop a Data Processing Plan: Map the required data elements to existing electronic health records and other data sources. Define a plan for extracting, cleaning, and aggregating this data from all sites into a central registry [86].
- Test Changes via PDSA Cycles: Use Plan-Do-Study-Act (PDSA) cycles to test small, iterative changes in clinical practice. Sites share their PDSA results and learnings with the entire collaborative.
- Implement & Measure: Implement successful changes from the PDSA cycles. Continue to collect data from the central registry to measure the impact on the primary and secondary outcomes.
- Analyze and Share Results: Analyze the aggregated data to assess the effectiveness of the intervention. Present findings at scientific meetings and publish the results to contribute to the broader evidence base [84].
Workflow Visualization
The following diagram illustrates the logical workflow for establishing and operating a multi-national pediatric research collaborative, integrating ethical frameworks and data management.
Conclusion
Ethical pediatric research consent is a dynamic and multifaceted process that requires balancing rigorous regulatory adherence with compassionate, child-centric communication. Key takeaways include the necessity of a dual consent/assent model, the critical role of minimizing coercion and therapeutic misconception, and the importance of adaptable strategies for diverse settings and cultures. Future efforts must focus on enhancing public engagement to build trust, developing standardized yet flexible international guidelines, and continuing to refine methodological best practices. By prioritizing these ethical foundations, the research community can advance pediatric healthcare while steadfastly protecting the rights and welfare of its youngest participants.
References
- 1. A Summary of Important Documents in the Field of Research ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2632196/]
- 2. Nuremberg Code | History, Date, & 10 Points [https://www.britannica.com/topic/Nuremberg-Code]
- 3. Reports, Declarations, Codes & Guidelines - Academics [https://academics.lmu.edu/irb/reportsdeclarationscodesguidelines/]
- 4. Ethics Challenges in Pediatric Research - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10715381/]
- 5. The creation of the Belmont Report and its effect on ethical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9700634/]
- 6. Ethical Principles - Human Research Protection Program [https://hrpp.usc.edu/policies/ethical-principles/]
- 7. The Belmont Report - Ethical Principles [https://www.washington.edu/research/hsd/guidance/ethical-principles/belmont/]
- 8. Ethical Challenges with the Informed Consent Process in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10928675/]
- 9. Ethical Aspects and Challenges in Pediatric Clinical Trials [https://www.linical.com/articles-research/ethical-aspects-and-challenges-in-pediatric-clinical-trials]
- 10. Informed Consent and Assent [https://www.chp.edu/research/research-excellence/researchers/informed-consent]
- 11. Children and bioethics: clarifying consent and assent in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10075240/]
- 12. GUIDELINE 17:RESEARCH INVOLVING CHILDREN AND ... [https://www.ncbi.nlm.nih.gov/books/NBK614432/]
- 13. Ethical Principles and Standards for Developmental ... [https://www.srcd.org/about-us/ethical-principles-and-standards-developmental-scientists]
- 14. IRB Guideline I - Parental Consent and Child Assent [https://researchsupport.psu.edu/orp/irb/irb-resources-training-and-events/irb-guidelines/irb-guideline-i-parental-consent-and-child-assent/]
- 15. Understanding Ethical Issues of Research Participation from the Perspective of Participating Children and Adolescents: A Systematic Review - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5724520/]
- 16. Children in Research - Pitt HRPO - University of Pittsburgh [https://www.hrpo.pitt.edu/children-research-0]
- 17. Department of Health and Human Services Policy for the ... [https://www.federalregister.gov/documents/2024/10/24/2024-24399/department-of-health-and-human-services-policy-for-the-protection-of-human-research-subjects-update]
- 18. Additional Protections for Children [https://www.fda.gov/science-research/clinical-trials-and-human-subject-protection/additional-protections-children]
- 19. Study Risk Levels | Rutgers Research [https://research.rutgers.edu/researcher-support/research-compliance/human-research-protection-program/irb-process/study-risk-levels]
- 20. Defining "Minimal Risk" in Clinical Research [https://cms.centerwatch.com/insights/defining-minimal-risk-in-clinical-research/]
- 21. Minimal Risk in Pediatric Research: A Philosophical Review ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5618101/]
- 22. Obtaining Consent from Both Parents for Pediatric Research [https://pmc.ncbi.nlm.nih.gov/articles/PMC3529951/]
- 23. Consent and assent in paediatric research in low-income ... [https://bmcmedethics.biomedcentral.com/articles/10.1186/1472-6939-15-22]
- 24. Drafting the Informed Consent Form - UCI Office of Research [https://research.uci.edu/human-research-protections/subject-enrollment/informed-consent/drafting-the-informed-consent-form/]
- 25. Informed Consent Guidelines & Templates [https://hrpp.umich.edu/irb-health-sciences-and-behavioral-sciences-hsbs/informed-consent-guidelines-templates/]
- 26. Tips for Writing Consent Forms - Division of Research [https://research.tamu.edu/research-compliance/human-research-protection-program/tips-for-writing-consent-forms/]
- 27. Boston Children's Research Informed Consent [https://research.childrenshospital.org/resources/institutional-review-board/information-researchers/informed-consent]
- 28. Consent Forms | Division of Research [https://division-research.brown.edu/research-cycle/conduct-research/human-subjects-research/submit-study/forms-templates/consent-forms]
- 29. Honoring childhood agency in developmental research [https://pmc.ncbi.nlm.nih.gov/articles/PMC12456739/]
- 30. Assent - Under Age 18 | Office of Human Research Protections [https://human.research.wvu.edu/guidance/informed-consent/assent-under-age-18]
- 31. The Assent Process with Minors | Guides & Resources [https://www.tc.columbia.edu/institutional-review-board/guides--resources/the-assent-process-with-minors/]
- 32. Implementing Ethical Practices for Young Children's Assent ... [https://www.mdpi.com/2227-7102/15/5/571]
- 33. Obtaining Parental Permission & Child Assent [https://clinicalresearch.unc.edu/playbooks/my-study-lifecycle/study-conduct/obtaining-parental-permission-and-child-assent/]
- 34. Elements must meet minimum color contrast ratio thresholds [https://dequeuniversity.com/rules/axe/4.8/color-contrast]
- 35. Text has minimum contrast[proposed] | ACT Rule | WAI [https://www.w3.org/WAI/standards-guidelines/act/rules/afw4f7/proposed/]
- 36. Key Considerations for Flowchart Accessibility - MN.gov [https://mn.gov/mnit/about-mnit/accessibility/news/?id=38-436349]
- 37. fillcolor [https://graphviz.org/docs/attrs/fillcolor/]
- 38. Text has enhanced contrast | ACT Rule | WAI [https://www.w3.org/WAI/standards-guidelines/act/rules/09o5cg/]
- 39. Pediatric Consent Errors | Applied Clinical Trials Online [https://www.appliedclinicaltrialsonline.com/view/pediatric-consent-errors]
- 40. Ethical Informed Consent, Child Ethics Involving Children [https://childethics.com/informed-consent/]
- 41. Ethical guidelines for human research on children and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11472871/]
- 42. Practical and Ethical Issues in Pediatric Clinical Trials [https://www.appliedclinicaltrialsonline.com/view/practical-and-ethical-issues-pediatric-clinical-trials]
- 43. Ongoing Consent & Re-Consent - One UNC Clinical Research [https://clinicalresearch.unc.edu/playbooks/my-study-lifecycle/study-conduct/ongoing-consent-and-re-consent/]
- 44. Re-Consent - Office for Human Subject Protection [https://www.rochester.edu/ohsp/resources/consent/re-consent/]
- 45. Relational ethics, informed consent, and informed assent in ... [https://pubmed.ncbi.nlm.nih.gov/35665498/]
- 46. Consent and assent in research on children [https://embassy.science/wiki/Theme:39868075-a4e2-4f56-829d-6ccfdc288bdf]
- 47. Consent and Assent for Research on Children [https://embassy.science/wiki/Theme:6feb603b-78db-43b6-9722-a8833ef0d8c7]
- 48. Targeting Persons with Known Interpersonal Relationships for Human Subjects Research | Policies | Brown University [https://policy.brown.edu/policy/targeting-interpersonal-relationships]
- 49. How IRBs view and make decisions about coercion and undue influence - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3604028/]
- 50. Ethical Issues in International Research in Pediatric Psychology [https://pmc.ncbi.nlm.nih.gov/articles/PMC12638410/]
- 51. Key Considerations for Conducting Clinical Trials in ... [https://clinixir.com/blog/key-considerations-for-conducting-clinical-trials-in-pediatrics]
- 52. The Therapeutic Misconception: A Threat to Valid Parental ... [https://pubmed.ncbi.nlm.nih.gov/18792535/]
- 53. hope, trust and misconception in paediatric research [https://pubmed.ncbi.nlm.nih.gov/22350619/]
- 54. 6 step guide: Successfully navigating complexities of drug ... [https://www.norstella.com/6-step-guide-successfully-navigating-complexities-drug-development/]
- 55. Opt-In and Opt-Out Consent Procedures for the Reuse of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10015347/]
- 56. Opt-In vs. Opt-Out Rights: Key Differences [https://www.accountablehq.com/post/opt-in-vs-opt-out-data-rights]
- 57. A user-driven consent platform for health data sharing in ... [https://www.nature.com/articles/s41746-025-02147-3]
- 58. Guidance: Electronic Informed Consent [https://research.lehigh.edu/policies-guidance-forms/guidance-electronic-informed-consent]
- 59. New report published on simplified arrangements for ... [https://www.hra.nhs.uk/about-us/news-updates/new-report-published-on-simplified-arrangements-for-consent-in-clinical-trials/]
- 60. Consent management challenges - and how to overcome ... [https://www.resourcespace.com/blog/consent-management-the-challenges-and-tips-to-overcome-them]
- 61. Overcoming language barriers in pediatric clinical research [https://www.sciencedirect.com/science/article/abs/pii/S2352552525001306]
- 62. Cultural considerations for informed consent in paediatric ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6307601/]
- 63. 'Some parts of the consent form are written using complex ... [https://bmcmedethics.biomedcentral.com/articles/10.1186/s12910-024-01147-4]
- 64. Guidelines for Developing Telehealth Services and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5048262/]
- 65. Informing Physician Strategies to Overcome Language ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5903268/]
- 66. Study examines how pediatric residents across Canada ... [https://www.bcchr.ca/news/study-examines-how-pediatric-residents-across-canada-use-interpreters-care-patients]
- 67. Overcoming language barriers in pediatric care: a multilingual ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10917955/]
- 68. Family Experience in a Regional Participant Contact ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9938699/]
- 69. Researchers Highlight Ethical Concerns When Clinical ... [https://www.bu.edu/sph/news/articles/2025/researchers-highlight-ethical-concerns-when-clinical-trials-are-cut-short/]
- 70. Ethical challenges in research involving children affected ... [https://www.frontiersin.org/journals/developmental-psychology/articles/10.3389/fdpys.2025.1654278/full]
- 71. Guardians and research staff experiences and views about ... [https://bmcmedethics.biomedcentral.com/articles/10.1186/s12910-022-00865-x]
- 72. Barriers and Facilitators to Obtaining Informed Consent in a ... [https://pubmed.ncbi.nlm.nih.gov/30866724/]
- 73. Utilization of family members to provide hospital care in Malawi [https://pmc.ncbi.nlm.nih.gov/articles/PMC3623026/]
- 74. Informed consent approaches for clinical trial participation of ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0237088]
- 75. Regulatory Framework for Protecting Child Participants ... - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK25558/]
- 76. Video consent is preferred over written informed consent in ... [https://journals.plos.org/digitalhealth/article?id=10.1371/journal.pdig.0001067]
- 77. Urban-Suburban Differences in Public Perspectives on ... [https://www.jmir.org/2025/1/e60324/]
- 78. Institutional Review Boards Frequently Asked Questions [https://www.fda.gov/regulatory-information/search-fda-guidance-documents/institutional-review-boards-frequently-asked-questions]
- 79. What is an IRB? The Role of Ethics Committees in Research [https://intuitionlabs.ai/articles/role-irb-ethics-committee]
- 80. Conducting Pediatric Studies: Addressing Ethical and ... [https://www.precisionformedicine.com/blog/pediatric-clinical-trials-challenges/]
- 81. Final Guidance Released on IRB Written Procedures [https://www.clinicalpathwaysresearch.com/blog/2025/6/3/final-guidance-released-on-irb-written-procedures]
- 82. Ethical Issues in International Research in Pediatric ... [https://link.springer.com/article/10.1007/s10880-025-10087-z]
- 83. Chapter: 5 Raising Awareness of Regulatory, Legal, and ... [https://www.nationalacademies.org/read/9816/chapter/7]
- 84. The Pediatric Experience Collaborative: Paving the Way in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12235225/]
- 85. Harmonizing Data Visualizations on Child Health and Well- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10749642/]
- 86. A structured approach for data collection and visualization ... [https://www.sciencedirect.com/science/article/pii/S2772610X25000273]
- 87. Multisector collaborations at children's hospitals to address ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12343070/]
- 88. Pediatric Collaborative Networks for Quality Improvement ... [https://www.sciencedirect.com/science/article/abs/pii/S1876285913002052]
- 89. Data visualization as an intervention for pediatric chronic pain [https://pilotfeasibilitystudies.biomedcentral.com/articles/10.1186/s40814-022-01170-5]